5 Foods That Are Silently Harming Your Egg Quality (And the Clinical Reason Why)
You Are Eating Well. So Why Is Nothing Working?
You have cut out alcohol. You have started taking a prenatal vitamin. You are going to bed earlier, drinking more water, and tracking your cycle with a precision that would impress an astronomer. You are doing everything right. And still, month after month, nothing is happening.
Or perhaps you have had a round of IVF, and the results came back with a note about “poor egg quality.” Nobody explained what caused it. Nobody gave you a roadmap for what to do differently. You were handed a protocol and sent home.
You typed “foods that destroy egg quality” into the search bar at half past midnight. You found a list. But nobody explained why. Nobody told you what is actually happening inside the follicle, at the cellular level, when you eat these things. Nobody gave you the clinical picture.
That is what this article is for.
Because here is the truth that does not get said loudly enough: what you eat directly affects the environment inside your ovarian follicles, the microscopic fluid-filled sacs where your eggs develop and mature. And some of the foods most commonly found in a modern woman’s diet are actively working against that environment, in ways that are biologically specific and clinically significant.
This is not about guilt. This is about information. And once you have it, you can use it.

What Egg Quality Actually Means, and Why Your Diet Is Central to It
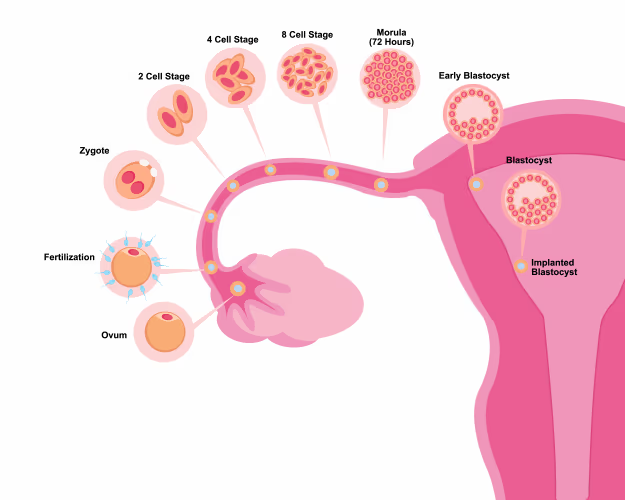
When fertility specialists talk about “egg quality,” they are referring to the chromosomal and mitochondrial health of the egg, the oocyte, at the moment of ovulation and fertilisation. A high-quality egg has the correct number of chromosomes (46), a functioning set of mitochondria (the energy-producing structures inside every cell), and a surrounding follicular environment that is low in oxidative stress and well-supplied with the nutrients the egg needs to mature and, if fertilised, develop into a healthy embryo.
Think of the follicle as a greenhouse. The egg is the seedling inside it. The quality of that greenhouse, its temperature, its chemical balance, its nutrient supply, determines how well the seedling develops. You can have the right seedling and still fail to grow a healthy plant if the environment is wrong. Diet shapes that environment profoundly.
Here is the featured snippet answer, because you deserve a clear one: foods that destroy egg quality are those that elevate oxidative stress (cellular damage caused by unstable molecules called free radicals), disrupt hormonal signalling (particularly oestrogen and insulin), or impair mitochondrial function in the developing egg. The five dietary categories most strongly linked to poor egg quality through clinical research are: ultra-processed foods high in refined carbohydrates; dietary trans fats; high-mercury fish and seafood; alcohol; and heavily pesticided produce that introduces endocrine-disrupting compounds into the body.
What makes this topic persistently underserved in mainstream medicine is that most fertility consultations focus on hormonal protocols and procedural timing, while the foundational role of diet in follicular health is covered in a single leaflet, if at all. The research is there. The clinical translation is not.
5 Foods That Destroy Egg Quality: The Clinical Mechanism Behind Each One
1. Ultra-Processed Foods and Refined Carbohydrates: The Insulin Disruption
This category includes white bread, packaged cereals, crisps, processed snack bars, instant noodles, sugary drinks, and the vast majority of convenience foods found on supermarket shelves. These are foods that have been industrially modified to the point where their original nutritional matrix, fibre, protein, micronutrients, has been largely removed, and where refined starches and added sugars dominate.
The mechanism by which these foods harm egg quality is both direct and indirect, and it begins with insulin.
When you eat refined carbohydrates, your blood glucose rises rapidly. Your pancreas responds by releasing insulin, a hormone whose job is to shuttle that glucose into cells. When this happens frequently and in large quantities, as it does in a diet high in processed foods, the cells begin to develop insulin resistance. They stop responding to insulin’s signal as efficiently. This forces the pancreas to produce even more insulin to achieve the same effect, and chronically elevated insulin levels have a cascade of reproductive consequences.
Elevated insulin stimulates the ovaries to produce androgens, including testosterone, at higher levels than normal. This androgenic environment interferes with follicular development and with the maturation of the egg itself. It disrupts the hormonal signalling that governs ovulation, which is why polycystic ovary syndrome (PCOS), a condition characterised by insulin resistance, is one of the leading causes of ovulatory dysfunction and reduced egg quality.
But there is more. Refined carbohydrates drive oxidative stress. This means they increase the production of reactive oxygen species (free radicals) inside the body’s cells. Free radicals cause damage to cell membranes, to DNA, and critically, to mitochondria. Since the mitochondria inside an egg cell are responsible for producing the energy required for fertilisation, cell division, and embryo development, mitochondrial damage in the oocyte translates directly into impaired egg function.
Research published in reproductive endocrinology literature has consistently linked dietary glycaemic load, that is, how dramatically a diet raises blood glucose overall, with poorer outcomes in IVF and natural conception cycles alike. Healthline’s comprehensive guide to egg quality and fertility notes that diets promoting insulin resistance are among the most well-evidenced dietary threats to female fertility.
The practical implication is this. You do not need to eliminate all carbohydrates. The goal is to shift the quality and source of the carbohydrates you eat, away from refined, stripped, rapidly digested ones and toward complex, whole-food sources that release glucose more slowly, support insulin sensitivity, and come packaged with the antioxidants and fibre that protect the follicular environment.
2. Dietary Trans Fats: The Cell Membrane Saboteur
Trans fats are a category of artificially altered fats created through a process called partial hydrogenation, in which liquid vegetable oils are chemically treated with hydrogen to make them solid and shelf-stable at room temperature. They are found in many commercially produced baked goods, fried fast foods, certain margarines, and any product whose ingredient label lists “partially hydrogenated oil.”
Many countries have significantly restricted or banned industrially produced trans fats in recent years, but they still appear in processed foods imported from regions with less stringent regulations, and they are still present in foods produced before regulatory changes came into effect. Natural trans fats also occur in small amounts in dairy and meat products, though these appear to behave differently from their industrial counterparts.
The mechanism by which industrial trans fats harm egg quality is elegant in its biology, and troubling in its implications.
Every cell in the body, including the oocyte and the cells lining the ovarian follicle, is surrounded by a membrane made primarily of phospholipids, a type of fat. The fluidity and integrity of that membrane determines how well the cell communicates with its environment, how efficiently it transports nutrients, and how responsive it is to hormonal signals. Healthy unsaturated fats, the kind found in olive oil, avocado, and nuts, maintain membrane fluidity and support optimal cell function.
Trans fats are incorporated directly into cell membranes in place of healthy fats. When they are, they make the membrane rigid and dysfunctional. In follicular cells, this rigidity impairs the cell’s ability to respond to LH (luteinising hormone), the hormonal signal that triggers ovulation and final egg maturation. It also reduces the cell’s ability to transport the antioxidants, nutrients, and signalling molecules that the maturing egg needs.
A landmark cohort study known as the Nurses’ Health Study II followed over 18,000 premenopausal women over an eight-year period and found a statistically significant association between higher intake of industrial trans fats and an increased risk of ovulatory infertility. Every two percent increase in energy intake from trans fats, when substituted for carbohydrates, was associated with a more than twofold greater risk of ovulatory infertility.
This is not a theoretical risk. It is a dose-dependent, biologically plausible, and well-documented one.
The practical point is to read ingredient labels with care, particularly on commercially baked goods and imported processed foods, and to replace trans-fat-containing cooking oils and spreads with whole-food fat sources: extra virgin olive oil, avocado oil, nuts, seeds, and oily fish.
3. High-Mercury Fish and Seafood: The Mitochondrial Threat
This is the entry that most fertility specialists flag, and with good reason. Fish is widely, and correctly, promoted as an excellent protein source for women trying to conceive. Oily fish in particular provides omega-3 fatty acids, specifically EPA and DHA (eicosapentaenoic acid and docosahexaenoic acid), which are anti-inflammatory, support hormonal balance, and are genuinely beneficial for egg quality.
The complication is mercury.
Mercury is a heavy metal that accumulates in aquatic ecosystems and concentrates in the fatty tissue of large, predatory fish. The higher up the food chain the fish sits, the more mercury it contains, a process called biomagnification. The fish with the highest mercury concentrations include swordfish, shark, king mackerel, tilefish, and bigeye tuna.
The mechanism by which mercury damages egg quality is through its direct toxicity to mitochondria. Mercury ions bind to sulphur-containing proteins in the mitochondria, disrupting the electron transport chain, the biochemical process by which mitochondria generate ATP, the cell’s primary energy currency.
For an oocyte, this matters enormously. The egg cell contains more mitochondria than almost any other cell in the human body, because the fertilisation process and the first several rounds of cell division that follow it are entirely dependent on the energy those mitochondria produce. Mitochondrial dysfunction in the egg translates directly into impaired fertilisation rates, arrested embryo development, and increased rates of chromosomal errors (aneuploidy) in the resulting embryo.
Research published in reproductive toxicology journals has demonstrated that women with higher blood mercury levels have significantly lower rates of fertilisation and blastocyst formation in IVF cycles compared to women with lower levels. Mercury also appears to concentrate in follicular fluid, the immediate environment of the developing egg, suggesting that its toxic effects occur at the site of egg maturation.
The NHS guidance on foods to avoid in pregnancy recommends limiting shark, swordfish, and marlin to no more than one portion per week even before pregnancy, for precisely this reason.
The practical point is not to avoid fish, which would be counterproductive, but to choose fish with a high omega-3 profile and a low mercury burden. Salmon, sardines, mackerel (Atlantic, not king), trout, anchovies, and herring are all excellent choices. Tinned light tuna has a lower mercury concentration than bigeye or yellowfin. Swordfish, shark, and king mackerel are the ones to set aside.
4. Alcohol: The Oestrogen Disruptor and Oxidative Stressor
Alcohol is worth addressing directly and without either alarm or the kind of vague reassurance that leaves women no better informed. The clinical picture on alcohol and egg quality is clear enough that it warrants honest explanation.
When you consume alcohol, your liver processes it through a two-step enzymatic reaction. The first step converts alcohol into acetaldehyde, a compound that is directly toxic to cells, including the cells of the ovarian follicle. The second step converts acetaldehyde into acetate, which is relatively harmless. The problem is that the first step generates large quantities of reactive oxygen species, free radicals, that cause oxidative damage throughout the body. Inside the ovarian follicle, this oxidative damage affects the mitochondria of the developing oocyte and the granulosa cells (the supporting cells that surround and nourish the egg).
Alcohol also disrupts oestrogen metabolism in a clinically significant way. The liver, already occupied with processing alcohol, reduces its capacity to metabolise and clear excess oestrogen from the bloodstream. This leads to elevated circulating oestrogen levels, which can suppress the hypothalamic-pituitary-ovarian axis, the hormonal communication system that governs follicular development and ovulation. In plain terms: drinking alcohol can disrupt the very hormonal sequence that determines whether ovulation occurs, and whether the egg that matures is healthy.
Research consistently supports an association between alcohol intake and reduced fertility. Studies in women undergoing IVF have found that even moderate alcohol consumption in the weeks preceding an egg retrieval cycle is associated with significantly lower numbers of eggs retrieved, lower fertilisation rates, and reduced embryo quality. The associations hold even when adjusted for other lifestyle factors.
As I’ve seen with many patients, this is one of the most difficult changes to make, not because of addiction, but because alcohol is so woven into social life that eliminating or significantly reducing it feels like an identity shift rather than a health decision. The most practical framing is this: the follicular maturation process takes approximately 90 days. The eggs that ovulate in any given cycle began their developmental journey three months earlier. Reducing alcohol consumption for a minimum of three months before a planned conception attempt, or before a fertility treatment cycle, gives those eggs the most supportive environment possible during their critical developmental window.
5. Heavily Pesticided Produce and the Endocrine Disruptor Problem
This is the entry that generates the most controversy, partly because the science is still evolving and partly because “eat more vegetables” is such a consistent public health message that the idea of vegetables being problematic feels counterintuitive. Let me be precise.
Vegetables and fruit are profoundly beneficial for fertility. This is not in dispute. The concern is not with produce itself. It is with the pesticide residues on certain types of conventionally grown produce, specifically organochlorine pesticides and certain organophosphates, which have been identified as endocrine-disrupting compounds (EDCs).
Endocrine-disrupting compounds are chemicals that interfere with the body’s hormonal signalling systems. They can mimic oestrogen (acting as xenoestrogens, meaning “foreign oestrogens”), block androgen receptors, interfere with thyroid hormone function, or directly disrupt the hormonal communication between the hypothalamus, pituitary, and ovaries.
Inside the ovarian follicle, EDC exposure has been linked to abnormal follicular development, reduced oocyte competence (the egg’s ability to be fertilised and develop normally), and an increased rate of chromosomal errors during the final maturation of the egg. Research published in Environmental Health Perspectives and in reproductive endocrinology journals has found measurable concentrations of organochlorine pesticide residues in follicular fluid, the immediate environment of the developing egg, in women undergoing IVF, and has associated higher concentrations with poorer embryological outcomes.
The produce most likely to carry high pesticide residues are strawberries, spinach, kale, peaches, pears, cherries, apples, grapes, bell peppers, hot peppers, blueberries, and green beans. These are sometimes referred to in nutritional research as “high-residue produce.”
The practical guidance here is not to stop eating vegetables. It is to prioritise organic versions of the highest-residue produce where budget allows, to wash all produce thoroughly with cold water, and to diversify the types of produce you eat rather than relying heavily on a small number of high-residue items. The irony is that many of the foods on the high-residue list are also nutritionally excellent. The goal is not elimination, but informed selection.
In My 19 Years of Clinical Practice, What I Have Seen Most Often Is…
In my 19 years of clinical practice, what I have seen most often is women who have done extraordinary amounts of research, who have overhauled their lifestyles in the most committed way, but who have been given one critical piece of advice too late. By the time a woman arrives at a fertility clinic and receives a diagnosis of diminished ovarian reserve or poor egg quality, she has usually spent months or years eating a diet that was technically “healthy” by mainstream standards but that was, in terms of follicular biology, working against her without her knowing it.
The most common pattern I see is the combination of high glycaemic eating with chronic low-grade alcohol intake. Both seem modest on their own. A bowl of pasta here, a few glasses of wine at the weekend. But their combined effect on oxidative stress inside the ovarian follicle, and on the hormonal environment in which the egg is maturing, is significantly greater than either factor alone.
What compounds the problem is the 90-day development window. Because eggs take three months to reach maturity, dietary changes made in the month before an IVF cycle or a natural conception attempt have limited impact on the eggs already partway through that cycle. The women who see the most meaningful improvements in egg quality metrics are those who make dietary changes early and sustain them across a minimum of three full months. The earlier the conversation about nutrition happens in a fertility journey, the better the outcomes tend to be. This is a gap in standard care that genuinely costs women their best chances.
When to See a Specialist: Red Flags and Timeframes
The following situations warrant specialist referral, and I would encourage you not to delay.
If you have been trying to conceive for 12 months without success and are under 35, request a referral to a reproductive endocrinologist. Ask your GP to arrange baseline fertility investigations: a day-2 or day-3 FSH and oestradiol, an AMH (anti-Müllerian hormone) level, and a transvaginal ultrasound for antral follicle count. These four tests together give a clear picture of your ovarian reserve.
If you are aged 35 or over and have been trying for six months without success, do not wait for the 12-month marker. Request the same tests at six months. Ovarian reserve declines with age, and earlier investigation preserves your options.
If you have been diagnosed with PCOS, endometriosis, or thyroid dysfunction, these conditions directly affect egg quality and require specialist input before conception, not after a year of trying. Book with a reproductive endocrinologist at the point of diagnosis if you are planning a pregnancy within the next two to three years.
If you have had two or more miscarriages, even early ones, this warrants investigation with a recurrent miscarriage specialist. Poor egg quality is one of the leading contributors to chromosomal errors in early embryos and subsequent miscarriage. A reproductive immunologist may also be relevant if immune factors are suspected.
If your AMH level comes back below 1.0 ng/mL, request an urgent consultation with a reproductive endocrinologist. Low AMH indicates reduced ovarian reserve, and the dietary window for supporting the remaining follicles becomes time-sensitive at this point.
What You Know Now Can Change What Happens Next
You came to this article looking for answers. You found them. And what you have now is something genuinely useful: a clear, clinically grounded understanding of how specific foods damage the environment in which your eggs develop, and precisely why that damage occurs.
The most important takeaway is the 90-day principle. The eggs ovulating today began their development three months ago. Which means that the most meaningful investment you can make in your egg quality is not what you do this week, but what you sustain over the next three months. Reducing refined carbohydrates, eliminating trans fats, choosing low-mercury fish, moderating alcohol, and prioritising lower-residue or organic produce: these are not perfectionistic demands. They are targeted, evidence-informed choices that create a measurably better environment for your eggs to mature in.
You do not need to be perfect. You need to be consistent.
Read Next: How to Build a Fertility-Supportive Meal Plan: A Clinician’s Guide to Eating for Egg Quality
Or drop a comment below and tell me where you are in your fertility journey. Every question matters.
This article is for informational purposes only and does not constitute medical advice. Always consult a qualified healthcare provider before making any changes to your health or treatment plan.