The Hidden Truth About 6 Hormonal Imbalance Symptoms Women Ignore Every Day
You’ve Been Telling Them Something Is Wrong
It’s 11pm on a Tuesday. You’re exhausted in a way that feels almost cellular, yet you’re lying awake with your mind skittering through nothing in particular. You gained four kilograms since last spring without changing anything meaningful in your diet. Your hair is coming out in soft, alarming clusters every time you shower. You snapped at your partner this morning over something small and felt a wave of shame that lingered for hours.
You’ve mentioned it to your GP. More than once.
You were told you’re probably stressed. You were told it could be your age. You were offered a mild antidepressant. You were told that blood tests came back “normal.” And each time, you walked out of that consulting room feeling unseen, slightly foolish for even bringing it up, and quietly certain that something real is being missed.
You are not imagining it.
What you may be experiencing is a constellation of hormonal imbalance symptoms that, when taken individually, are easy to dismiss and, when taken together, tell a coherent clinical story. Hormonal imbalance symptoms in women are among the most reported and least systematically investigated complaints in primary care. The problem is not that medicine doesn’t know these symptoms exist. The problem is that medicine too often meets them one at a time, in silos, without the connective clinical thinking that would reveal what is actually driving them.
This article is designed to give you that connective thinking.
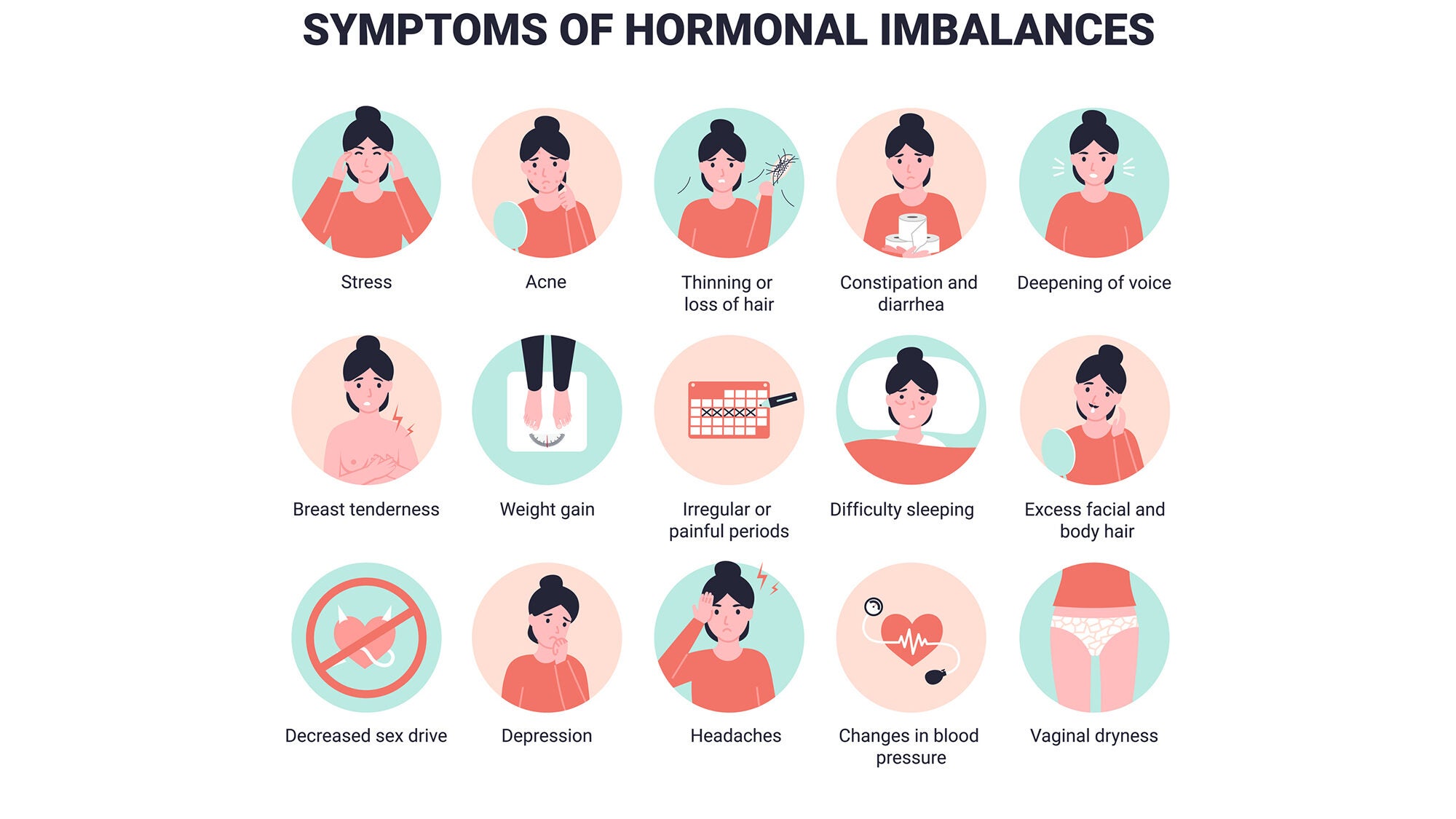
What Hormonal Imbalance Actually Means
The phrase “hormonal imbalance” is used so widely and so loosely that it has almost lost clinical meaning in popular conversation. But in a medical context, it refers to something precise and measurable: a disruption in the levels, ratios, or rhythms of one or more hormones in your endocrine system, the network of glands and organs responsible for producing, transporting, and regulating the chemical messengers that govern virtually every function in your body.
Think of your hormones as an internal postal system. Oestrogen (the primary female sex hormone, produced mainly in the ovaries) delivers messages to your brain, bones, cardiovascular tissue, and skin. Progesterone (produced after ovulation, during the second half of your menstrual cycle) carries instructions to your nervous system, your uterine lining, and your sleep architecture. Cortisol (produced by the adrenal glands in response to stress) speaks to your metabolism, immune function, and blood sugar regulation. Thyroid hormones, produced by the butterfly-shaped thyroid gland at the base of your throat, reach every single cell in your body and govern your metabolic rate, energy production, and body temperature.
When one part of this postal system sends the wrong message, sends it too frequently, or goes quiet altogether, the whole body feels it. That is why hormonal imbalance rarely presents as a single, clean complaint. It presents as a cluster: fatigue here, mood disruption there, weight changes, brain fog, disturbed sleep, skin and hair changes. Nothing necessarily catastrophic on its own. Everything significant when viewed together.
What hormonal imbalance means in plain terms: Hormonal imbalance occurs when the levels or rhythms of one or more key hormones in your body deviate from what your systems need to function optimally. In women, the most commonly implicated hormones are oestrogen, progesterone, cortisol, insulin, and thyroid hormones. Symptoms can range from persistent fatigue and cognitive difficulty to irregular periods, mood disorders, and unexplained changes in weight, hair, and skin.
The reason this area is so poorly served by mainstream medicine is structural. Standard blood panels typically test hormone levels at a single point in time, often at an arbitrary moment in your menstrual cycle, missing the dynamic fluctuations and cross-hormonal relationships that define hormonal health across the cycle and across the decades of a woman’s reproductive life.
You deserve an assessment that matches the complexity of what you are experiencing.
The Five Hormones Behind Most of What You’re Feeling
Before looking at the specific symptoms, it is worth understanding which hormones are most commonly involved and why. These five are the most frequent drivers of hormonal imbalance symptoms in women of reproductive age and beyond.
Oestrogen
Oestrogen is produced primarily in the ovaries, with smaller contributions from the adrenal glands and fat tissue. It governs the menstrual cycle, supports bone density, regulates mood, protects cardiovascular health, maintains skin collagen, and modulates cognitive function. Oestrogen levels fluctuate throughout the menstrual cycle and decline progressively from perimenopause onward. Disruptions to oestrogen, whether too high, too low, or poorly proportioned relative to progesterone, are implicated in a wide range of symptoms including mood changes, cognitive difficulty, sleep disruption, and skin changes.
Progesterone
Often overlooked in favour of oestrogen, progesterone is the calming counterbalance to oestrogen’s stimulating effects. Produced by the corpus luteum (the temporary glandular structure formed in the ovary after ovulation) and, in smaller amounts, by the adrenal glands, progesterone has a direct sedative effect on the nervous system through its influence on GABA receptors. Low progesterone, whether from infrequent ovulation, chronic stress, or hormonal transition, is a key driver of anxiety, sleep disruption, and premenstrual mood symptoms.
Cortisol
Cortisol is your primary stress hormone, released by the adrenal glands in response to physical or psychological pressure. In short bursts, it is adaptive and essential. In the context of chronic, sustained stress, however, elevated cortisol disrupts thyroid function, impairs insulin sensitivity, promotes abdominal fat storage, and gradually depletes the building blocks needed to produce sex hormones. Cortisol dysregulation is one of the most common and least investigated contributors to the complex of symptoms many women attribute to “just being stressed.”
Thyroid Hormones
The thyroid produces triiodothyronine (T3) and thyroxine (T4), hormones that regulate the metabolic rate of every cell in the body. Hypothyroidism (underactive thyroid), in which insufficient thyroid hormone is produced, can cause profound fatigue, weight gain, hair thinning, brain fog, low mood, constipation, and cold intolerance. According to the NHS, thyroid disease is significantly more common in women than men, and many women live with subclinical thyroid dysfunction for years before receiving a diagnosis.
Insulin
Insulin, produced by the pancreas, regulates blood sugar by facilitating glucose uptake into cells. When insulin signalling is impaired, a condition known as insulin resistance, blood sugar remains elevated, fat storage increases (particularly in the abdominal area), energy levels fluctuate dramatically, and the environment becomes one that promotes androgen excess. Insulin resistance is a central feature of polycystic ovary syndrome (PCOS) and is increasingly recognised as a driver of hormonal disruption in women without a formal PCOS diagnosis.
Understanding these five hormonal players gives you the foundation to make sense of the symptoms that follow.
6 Hormonal Imbalance Symptoms Women Ignore Every Day
1. Fatigue That Sleep Simply Does Not Fix
This is perhaps the most universal hormonal imbalance symptom in women, and it is also the most consistently minimised.
You sleep seven or eight hours and wake feeling as though you have not slept at all. By mid-morning you are functioning on caffeine and effort alone. By early afternoon, you feel a specific, dense heaviness that is nothing like ordinary tiredness. It is a tiredness that sits in your muscles and behind your eyes simultaneously. You have adjusted your bedtime, reduced your alcohol intake, taken supplements, exercised more, exercised less. Nothing shifts it.
This pattern of unrefreshing sleep and persistent daytime fatigue is a hallmark of several distinct hormonal disruptions, each with its own clinical mechanism.
Thyroid insufficiency is the first to consider. When your thyroid gland is underproducing T3 and T4, your metabolic rate slows across all tissues, including the brain and musculoskeletal system. You feel the energetic effect of this cellular slowing as fatigue that is qualitatively different from the tiredness produced by a late night. Thyroid-related fatigue tends to be heavy and persistent, often accompanied by a generalised slowness in thought and movement.
Oestrogen decline is the second mechanism. Oestrogen supports the production of serotonin, a neurotransmitter involved in wakefulness, mood regulation, and sleep quality. It also plays a direct role in regulating your circadian rhythm, your body’s internal clock. As oestrogen levels fall, whether cyclically in the premenstrual phase, postpartum, or progressively through perimenopause, sleep architecture can fracture. You may fall asleep without difficulty but surface repeatedly from deep sleep, or wake in the early hours and find yourself unable to return to restorative sleep.
Cortisol dysregulation is the third and perhaps most commonly overlooked mechanism. Under conditions of chronic stress, whether physical, psychological, or both, your adrenal glands maintain elevated cortisol output over extended periods. This chronic elevation is initially stimulating, creating the false impression of capability. Over time, however, sustained high cortisol suppresses thyroid hormone conversion (the process by which the less active T4 is converted to the more metabolically active T3) and depletes the precursor hormones needed to produce progesterone and oestrogen. The result is a fatigue that seems to worsen the harder you push through it.
If your fatigue has persisted for more than six weeks, is accompanied by any other symptoms on this list, and is unresponsive to ordinary rest, it is not a consequence of modern life. It is a clinical signal. Investigate it.
2. Brain Fog, Memory Lapses, and Difficulty Concentrating
You reach for a word and it is not there. You read a paragraph twice, three times, and cannot retain it. You walk into a room and stand momentarily blank, unable to reconstruct your intention in being there. You notice that the sharpness you relied on in your professional and personal life seems to have acquired a film, a soft blurring of precision that no amount of focus seems to penetrate.
This cognitive experience has a name in clinical literature. It is often referred to as oestrogen-related cognitive fluctuation, and it is real, measurable, and mechanistically understood.
Oestrogen has direct neuroprotective effects on the brain. It supports acetylcholine, a neurotransmitter critical to memory formation, learning, and attention. It promotes neuroplasticity, the brain’s ongoing ability to form, strengthen, and reorganise neural connections. It modulates the activity of the prefrontal cortex, the region responsible for executive function, working memory, and complex decision-making. Neuroimaging research has demonstrated observable differences in brain metabolism during periods of significant oestrogen depletion, including the late luteal phase of the menstrual cycle, the postpartum period, and the perimenopausal transition.
This is not a subtle or subjective effect. In functional brain imaging studies, women in oestrogen-depleted states show reduced glucose metabolism in regions associated with memory and verbal fluency. Your experience of reaching for a word and finding only air has a neurobiological correlate that is entirely legitimate.
Thyroid hormone deficiency produces an almost indistinguishable pattern of cognitive impairment. Thyroid hormones regulate cerebral blood flow and the speed of neuronal signal transmission. A sluggish thyroid means slower neural processing, reduced working memory, and a generalised cognitive blunting that is often described by patients as feeling mentally “waded through.”
Cortisol excess, conversely, also impairs cognition, through a different mechanism. Chronically elevated cortisol promotes neuroinflammation, disrupts hippocampal neurogenesis (the ongoing production of new neurons in the memory-forming hippocampus), and degrades the precision of the prefrontal cortex. Women who have been under sustained, prolonged stress for months or years often notice a progressive decline in cognitive sharpness that they incorrectly attribute to ageing.
The important clinical message is this: brain fog is not a personality trait, a function of busyness, or an inevitable feature of getting older. It is a symptom. And like all symptoms, it has an underlying cause that is clinically accessible once the right questions are asked.
3. Unexplained Weight Gain, Particularly Around the Abdomen
This is the symptom that most frequently meets with the frustrating and reductive response: “eat less, move more.” For many women experiencing hormonally driven weight changes, that advice is not only unhelpful but mechanistically irrelevant.
You have not changed your diet significantly. Your activity levels are comparable to what they have always been. And yet, weight is accumulating specifically around your middle, a soft thickening at the waist that feels unlike the weight changes you experienced in your twenties. It resists the usual interventions. It seems to follow its own logic.
That logic is hormonal.
Cortisol is the primary driver of central, visceral weight gain (fat accumulation around the abdominal organs). When cortisol is chronically elevated, it activates lipoprotein lipase, an enzyme that promotes fat storage specifically in visceral adipose tissue (the fat that surrounds your internal organs). This is an evolutionary mechanism. Your adrenal glands, interpreting prolonged stress as a signal of environmental danger, prioritise fat storage in accessible, rapidly mobilisable locations. Your body, in other words, is doing exactly what it was designed to do. It is simply doing it in a contemporary environment where the stressor is a demanding job or a difficult relationship rather than a predator or a famine.
Insulin resistance compounds this significantly. When your cells become less responsive to insulin’s signals, glucose accumulates in the bloodstream rather than being absorbed for energy use. The pancreas responds by producing more insulin. Elevated circulating insulin then promotes further fat storage, particularly in the abdominal region, while simultaneously making it harder for the body to access stored fat as fuel. You feel hungry more frequently, particularly for carbohydrate-dense foods, because your cells are genuinely energy-deficient despite adequate caloric intake. The standard dietary advice of “eat less” is profoundly unhelpful here because the problem is not the quantity of food but the cellular response to it.
Oestrogen decline during perimenopause shifts fat distribution from the hips and thighs, where oestrogen promotes fat storage during reproductive years, to the abdomen. This redistribution is hormonally driven and occurs independently of caloric intake or exercise habits. It is a direct consequence of the changing oestrogen-to-androgen ratio that characterises the perimenopausal transition. Research published and compiled by Healthline’s clinical review process confirms that hormonal shifts in perimenopause directly alter fat distribution patterns in women, independent of lifestyle factors.
Thyroid insufficiency slows metabolic rate globally, meaning that the same caloric intake produces a greater surplus relative to a slower energetic expenditure. Weight gain in hypothyroidism tends to be modest (typically three to five kilograms) and is accompanied by fluid retention that further affects body composition measures.
If you are gaining weight specifically around the abdomen, and standard lifestyle adjustments are producing no measurable change, a full hormonal and metabolic assessment, including fasting insulin, cortisol, thyroid function, and sex hormones, is a reasonable and warranted clinical request.
4. Cyclical Mood Shifts, Anxiety, and Low Mood
This symptom is perhaps the most frequently dismissed, and the dismissal carries the heaviest personal cost.
You have been told you are emotional. You have been told everyone feels low sometimes. You have been offered a prescription for antidepressants. And through all of it, you have privately observed that your mood shifts follow a pattern, a predictable rhythm tied to your menstrual cycle, that no amount of “stress management” seems to alter.
If your mood changes are cyclical, recurring reliably in the week before your period and resolving within a day or two of bleeding beginning, this is not a generalised mood disorder. This is a hormonally mediated neurochemical event with a specific clinical name: premenstrual dysphoric disorder (PMDD), or its less severe but still clinically significant counterpart, premenstrual syndrome (PMS).
The mechanism involves the interaction between progesterone metabolism and the GABA receptor system in the brain. In the luteal phase (the two weeks between ovulation and menstruation), progesterone rises and then falls sharply. As progesterone is metabolised, it produces a compound called allopregnanolone, which in most women has a calming, anxiolytic (anxiety-reducing) effect on the nervous system through GABA receptor activation. In women who develop PMDD, the nervous system paradoxically becomes dysregulated in response to allopregnanolone fluctuations, experiencing its natural rise and fall as destabilising rather than calming. This is not a psychological sensitivity. It is a neurological one.
Separately, the oestrogen decline that occurs in the late luteal phase reduces serotonin synthesis and receptor sensitivity, contributing directly to low mood, irritability, and tearfulness in the days before menstruation.
As I’ve seen with many patients, the most important clinical shift in understanding cyclical mood disruption is recognising that the primary problem is not depression or anxiety as standalone diagnoses. It is hormonal sensitivity driving a neurochemical pattern. Treating it with a selective serotonin reuptake inhibitor (SSRI) may provide partial symptom relief, but it does not address the underlying hormonal dynamic, and it frequently does not address the symptom adequately.
Postpartum mood disruption carries the same fundamental mechanism at a much larger scale. After delivery, oestrogen and progesterone levels fall more steeply and more rapidly than at almost any other point in the female lifespan. This hormonal cliff is a direct contributor to the emotional vulnerability and instability of the postpartum period. The fall in oestrogen alone significantly disrupts serotonin, dopamine, and noradrenaline signalling simultaneously. Add sleep deprivation, physical recovery demands, and the psychological adjustment of early parenthood, and the neurochemical environment becomes acutely vulnerable.
If your mood changes are predictably cyclical, worsening in the premenstrual phase and resolving with menstruation, or if they emerged or intensified following childbirth, this pattern warrants specialist investigation rather than a generalised psychiatric response.
5. Sleep Disruption, Particularly Waking Between 2am and 4am
If you are waking in the early hours of the morning, alert and unable to return to sleep, with a mind that feels activated rather than sleepy, you may already be familiar with the particular quality of exhaustion this produces. It is not the pleasant heaviness of deep, restorative tiredness. It is a buzzing, restless, frustrated wakefulness at the hour when the world outside is entirely quiet and you cannot understand why your body is refusing to rest.
This specific pattern, early morning waking between 2am and 4am, is a recognised clinical feature of hormonal disruption, and it has several intersecting mechanisms.
The first involves cortisol rhythm. In a healthy circadian pattern, cortisol should reach its lowest point between midnight and 2am, then begin rising gradually in preparation for waking around 6am. In women with cortisol dysregulation, whether driven by chronic stress, adrenal overactivation, or blood sugar instability, cortisol can spike abnormally in the early morning hours, sometimes as early as 2am. This premature cortisol surge activates the sympathetic nervous system (your “fight or flight” system), raising heart rate, alerting the brain, and making return to sleep profoundly difficult.
Blood sugar dysregulation intersects with this mechanism directly. When blood sugar drops overnight, whether due to an imbalanced evening meal, insulin resistance, or reactive hypoglycaemia (a condition in which blood sugar falls sharply a few hours after eating), the body releases cortisol and adrenaline to raise blood glucose to safe levels. This emergency metabolic response is physiologically necessary but neurologically activating. The result is abrupt waking, often with a racing heart or mild anxiety, in the early hours.
Progesterone deficiency is the second key mechanism. Progesterone’s calming effect on the nervous system works in part through its metabolite allopregnanolone, which enhances the sensitivity of GABA receptors in the brain, effectively quieting the nervous system for deep, uninterrupted sleep. When progesterone is insufficient, whether in the late luteal phase, during perimenopause, or postpartum, this GABAergic sedation is reduced. The nervous system becomes more easily aroused in the lighter stages of sleep, producing fragmented nights even without full waking.
Oestrogen fluctuation is the third mechanism, manifesting most visibly as night sweats. Hot flushes and night sweats are driven by oestrogen’s role in regulating the hypothalamic thermostat, the brain structure responsible for maintaining body temperature. When oestrogen fluctuates erratically, as it does in perimenopause (which can begin in the mid to late thirties, well before most women expect it), the hypothalamic thermostat becomes unstable. Body temperature spikes briefly, triggering a sweating response that may or may not fully wake you but consistently disrupts the deeper, restorative stages of sleep.
The cumulative effect of these mechanisms is a pattern of persistently unrefreshing sleep that is distinct from insomnia in its classical form. If you are falling asleep easily but waking consistently between 2am and 4am, particularly alongside other symptoms on this list, this is a clinical pattern that deserves formal hormonal investigation rather than a prescription for sleep medication as a first response.
6. Hair Thinning, Adult Acne, and Changes in Skin Texture
Your hair, once thick, is now coming away in the shower in amounts that concern you. The density at your temples and across the crown feels different. In photographs you can see it. You are also, somehow, in your thirties or forties, dealing with acne, not the scattered kind of your teenage years but a specific, recurring pattern along the jawline and chin that arrives predictably before your period. And your skin has changed in a way that moisturiser cannot adequately address: drier, thinner, less resilient than it once was.
Each of these changes reflects a specific hormonal mechanism, and all three are clinically significant.
Hair thinning in women of reproductive and perimenopausal age most commonly involves androgens. Androgens are male hormones (including testosterone and dihydrotestosterone, or DHT) that are present in all women but become problematic when elevated in absolute terms or when hair follicles become overly sensitive to normal circulating levels. DHT binds to androgen receptors in hair follicles, progressively miniaturising them and shortening the active growth phase of the hair cycle. The result is a gradual, diffuse thinning across the scalp, often most visible at the temples and crown, that is called androgenic alopecia.
This pattern is most strongly associated with PCOS but is also seen in women without PCOS, particularly during perimenopause when declining oestrogen reduces its counterbalancing effect on androgens. Thyroid dysfunction, both hypothyroidism and hyperthyroidism, is an independent cause of diffuse hair loss in women, through a different mechanism involving the disruption of the hair growth cycle at the follicular level. Postpartum hair shedding (telogen effluvium) is driven by the abrupt hormonal shift following delivery and typically resolves within six to twelve months, though persistent postpartum hair loss beyond this window warrants a thyroid assessment.
Adult acne along the jawline and chin is one of the clearest cutaneous (skin-related) markers of androgen excess. Androgens stimulate sebaceous glands (the oil-producing glands embedded in the skin) to increase sebum production. Excess sebum creates a microenvironment within pores that promotes the proliferation of Cutibacterium acnes (the bacteria principally responsible for inflammatory acne lesions). This pattern, concentrated in the lower face rather than the forehead and nose typical of adolescent acne, and worsening cyclically in the premenstrual phase, is clinically distinguishable from acne driven by hygiene, diet, or stress alone.
Elevated insulin levels amplify androgen-driven acne by stimulating insulin-like growth factor 1 (IGF-1), which further increases sebum production and skin cell turnover in ways that promote pore blockage. This explains why adult acne frequently improves when insulin resistance is addressed, even before androgen levels are directly normalised.
Skin texture changes, including dryness, loss of suppleness, and reduction in elasticity, are closely linked to declining oestrogen. Oestrogen stimulates fibroblasts (cells in the dermis responsible for producing collagen, elastin, and hyaluronic acid) to maintain the structural integrity and hydration of skin tissue. As oestrogen declines, collagen synthesis slows, the dermis thins, and the skin’s capacity for water retention diminishes. This is not simply ageing. It is a specific, hormone-driven biological process. It begins earlier than most women expect, often in the late thirties, and accelerates significantly in the early perimenopausal years.
If you are experiencing any combination of hair thinning, jawline acne, and skin texture changes that have emerged or worsened over the past one to two years, particularly alongside irregular periods, fatigue, or weight changes, a full hormonal assessment is clinically appropriate and entirely reasonable to request.
What Comprehensive Hormonal Testing Should Actually Include
One of the most common frustrations women share in hormonal health consultations is being told their blood tests are “normal” despite experiencing clear and persistent symptoms. Understanding what comprehensive hormonal testing actually entails, as opposed to the limited panels often run in primary care, allows you to have a more informed and productive conversation with your GP or specialist.
A Full Thyroid Panel
Standard primary care testing often includes only TSH (thyroid-stimulating hormone), a pituitary hormone that signals the thyroid. TSH alone is an incomplete picture. A comprehensive thyroid assessment should include free T3, free T4, reverse T3, and thyroid antibodies (specifically TPO antibodies and Tg antibodies). Elevated thyroid antibodies, in the presence of normal TSH, can indicate the early stages of autoimmune thyroid disease (Hashimoto’s thyroiditis), which is one of the most common causes of subclinical hypothyroid symptoms in women.
A Full Sex Hormone Panel
A meaningful sex hormone assessment includes oestradiol (the primary circulating form of oestrogen), progesterone, total and free testosterone, DHEA-S (a precursor androgen produced by the adrenal glands), sex hormone binding globulin (SHBG), and FSH (follicle-stimulating hormone). Importantly, oestradiol and progesterone should be tested at specific, defined points in the menstrual cycle: oestradiol on cycle day 2 or 3, and progesterone seven days after confirmed ovulation. Testing these at arbitrary points in the cycle produces results that are difficult to interpret meaningfully.
Fasting Insulin and Blood Sugar
A fasting glucose test alone does not reveal insulin resistance. A fasting insulin test, run alongside fasting glucose, allows calculation of the HOMA-IR index, a reliable marker of insulin sensitivity. Elevated fasting insulin in the presence of normal fasting glucose is a common early marker of insulin resistance that is frequently missed because fasting glucose has not yet been affected.
Cortisol Rhythm Assessment
A single cortisol blood test gives a snapshot of cortisol at one moment in time. Since cortisol follows a diurnal rhythm (high in the morning, low by evening), a single measurement is often clinically uninformative. A four-point salivary cortisol test, which measures cortisol at four separate time points across the day, provides a far more useful picture of cortisol rhythm, adrenal function, and any disruption to the natural daily pattern.
Understanding these testing options empowers you to ask specific, informed questions at your GP appointment and to seek a referral when the initial assessment is insufficient to explain your symptoms.
The Clinical Insight: What I See Most Often
In my 19 years of clinical practice, what I’ve seen most often is a pattern I have come to call the dismissed constellation. A woman sits in my consulting room with a history that spans years and multiple practitioners. She has been prescribed an antidepressant for cyclical low mood. She has been advised to exercise more for fatigue that exercising only deepens. She has been told her hair loss is “just stress” and her adult acne is “probably dietary.” She has been given the oral contraceptive pill to regulate her periods, without any investigation into why her periods became irregular in the first place.
Each symptom has been responded to individually. Each response has, at best, partially addressed the specific complaint. And the underlying hormonal thread connecting all of them, perhaps an under-recognised transition into perimenopause, or an undiagnosed thyroid condition, or insulin resistance driving androgen excess, has never been identified.
This is not a criticism of any individual practitioner. It is a structural consequence of the way primary care is organised. A ten-minute appointment is not designed to identify a hormonal pattern that has developed over five or six years and presents across six different symptoms. The system defaults to symptomatic management because that is what is logistically achievable.
What changes outcomes for women in this situation is not a single test or a single prescription. It is arriving at a consultation with a coherent, documented account of the full symptom picture: when each symptom began, how it has changed over time, whether it follows a cyclical pattern, and what, if anything, has temporarily improved or worsened it. That documented account is the map that a skilled clinician can read to find the underlying territory.
When to Book a Specialist Consultation
Not every hormonal symptom warrants emergency action. Many are responsive to thorough, evidence-based assessment and targeted intervention over time. But certain symptom combinations or trajectories indicate that a primary care response is no longer sufficient and that specialist involvement is clinically appropriate.
Persistent, unrefreshing fatigue lasting more than six weeks despite adequate sleep, with no obvious lifestyle cause, warrants a full thyroid panel including antibodies. If TSH is borderline high (above 2.5 in the context of clear symptoms) or if thyroid antibodies are elevated, request a referral to an endocrinologist rather than accepting a “normal range” dismissal without clinical context.
Cyclical mood symptoms that predictably worsen in the seven to ten days before menstruation and resolve within two days of bleeding beginning, particularly if they are significantly impairing your ability to function, work, or maintain relationships, warrant a referral to a gynaecologist or reproductive endocrinologist with experience in PMDD. A general antidepressant prescription is not an adequate first-line response to a cyclical, hormonally driven condition.
Hair thinning alongside jawline acne and irregular periods, particularly if your periods are widely spaced (more than five weeks apart) or very heavy, warrants a full androgen panel and formal PCOS assessment by a gynaecologist. Do not accept “eat better and lose weight” as an explanation for this symptom cluster without a proper diagnostic evaluation.
Postpartum symptoms lasting beyond four months, including persistent fatigue, ongoing hair shedding, low mood or anxiety, and cognitive difficulty, should prompt a specific request for a full thyroid panel including TPO antibodies, to exclude postpartum thyroiditis. This condition affects up to 10 percent of postpartum women and is routinely missed because its symptoms are attributed to the demands of new parenthood.
Night sweats, early morning waking, and mood shifts that appear in your late thirties or early forties, even in the continued presence of regular menstrual cycles, may indicate the early stages of perimenopause. A gynaecologist can guide an appropriate hormonal assessment. You do not need to be in your late forties to have this conversation.
You Are Not Overreacting. You Are Under-Investigated.
The symptoms you have been living with, perhaps for months or years, are real. They are interconnected. And in the overwhelming majority of cases, they are addressable once the underlying hormonal picture is properly understood and properly assessed.
The single most important thing you can take from this article is this: your symptoms are not separate inconveniences to be managed in isolation. They are a conversation your body is having with you, consistently, in the same language, about the same underlying disruption. Learning to hear that conversation as a whole, rather than as a series of unrelated complaints, is what changes the clinical outcome.
Document your symptoms. Note their pattern. Record when they began, whether they cycle with your menstrual rhythm, and whether they have intensified following specific life events such as pregnancy, a period of prolonged stress, or a change in contraception. Take that documented picture to a specialist who is trained to read it.
You deserve care that meets the complexity of what you are experiencing.
Read Next: [How to Read Your Hormone Test Results: What Your GP Isn’t Telling You] or share this article with a woman in your life who has been told her symptoms are “just stress.” She may need this more than she knows.
This article is for informational purposes only and does not constitute medical advice. Always consult a qualified healthcare provider before making any changes to your health or treatment plan.
.