8 Gynaecologist-Approved Secrets to Maintaining Perfect Vaginal pH Balance That Prevent 90% of Intimate Infections
You’ve Done Everything Right. So Why Does It Keep Coming Back?
You showered this morning. You switched to cotton underwear months ago. You stopped using the scented wash you used to love, even though you miss the smell. You’ve read the forums, followed the advice, and taken the antibiotics. Twice. Maybe three times this year.
And yet, here you are again. That familiar, unsettling discomfort has returned. The subtle itch, the slightly unfamiliar discharge, the smell that you can’t quite describe but that you recognise immediately. It’s back, and you’re exhausted by it.
If this pattern sounds like yours, you are not imagining it. You’re not failing at self-care. And you are absolutely not alone. Millions of women experience exactly this cycle of infection, treatment, and recurrence. The reason it keeps returning is rarely what they’ve been told.
In most cases, the missing piece of the puzzle is something called vaginal pH balance. It’s a term your GP may have mentioned in passing, if at all, and it is one of the most critically underexplained aspects of women’s intimate health. Understanding it doesn’t require a medical degree. But it can genuinely change everything.
This is not a scare piece. There are no warnings here about what your body is doing wrong. This is a clinical guide, written in plain language, about how your body is designed to protect itself and exactly what you can do to support that system when life throws it off course.
What Vaginal pH Balance Actually Is (And Why It Matters More Than You’ve Been Told)
Vaginal pH balance refers to the level of acidity inside the vaginal canal. pH is measured on a scale from 0 to 14. A lower number means more acidic; a higher number means more alkaline. A healthy vaginal pH sits between 3.8 and 4.5, making it approximately as acidic as a glass of tomato juice or a ripe orange. That comparison is not random. It helps explain why this internal environment is so inhospitable to most of the bacteria and fungi that cause infections.
Think of your vaginal ecosystem as a well-tended garden. The acid is the soil quality. When the pH is right, the beneficial plants (in this case, Lactobacillus bacteria) thrive and crowd out the weeds. When the soil shifts, the weeds take over quickly. Even a small disruption to the pH, moving from 4.2 to 5.0, can tip the balance dramatically.
The vagina maintains its acidity primarily through a group of beneficial bacteria called Lactobacillus. These microorganisms produce lactic acid as a natural by-product of their metabolism. That lactic acid keeps the pH low, and the low pH keeps harmful pathogens from establishing themselves. It is a self-sustaining protective system that, when functioning well, most women never notice.
Here is the answer that many women search for but rarely find explained clearly: vaginal pH balance is the measure of acidity in the vaginal environment, and maintaining it within the healthy range of 3.8 to 4.5 is your body’s primary biological defence against bacterial vaginosis, yeast infections, and a range of sexually transmitted infections. A disruption in this range, even a modest one, creates a window of vulnerability that opportunistic organisms exploit rapidly.
What mainstream medicine consistently underserves is this: vaginal pH is not a fixed number. It shifts throughout your menstrual cycle, responds to sexual activity, medications, stress, diet, and even sleep quality. Understanding these fluctuations is not optional extra knowledge. It is foundational. And most women are never given it.
You can find further clinical context on how vaginal pH interacts with common infections in resources like Mayo Clinic’s complete guide to bacterial vaginosis, which outlines clearly how pH disruption underpins the most common intimate infection affecting women of reproductive age.
8 Evidence-Based Secrets to Maintain Healthy Vaginal pH Balance
Secret 1: Test Your Vaginal pH Before Assuming You Have an Infection
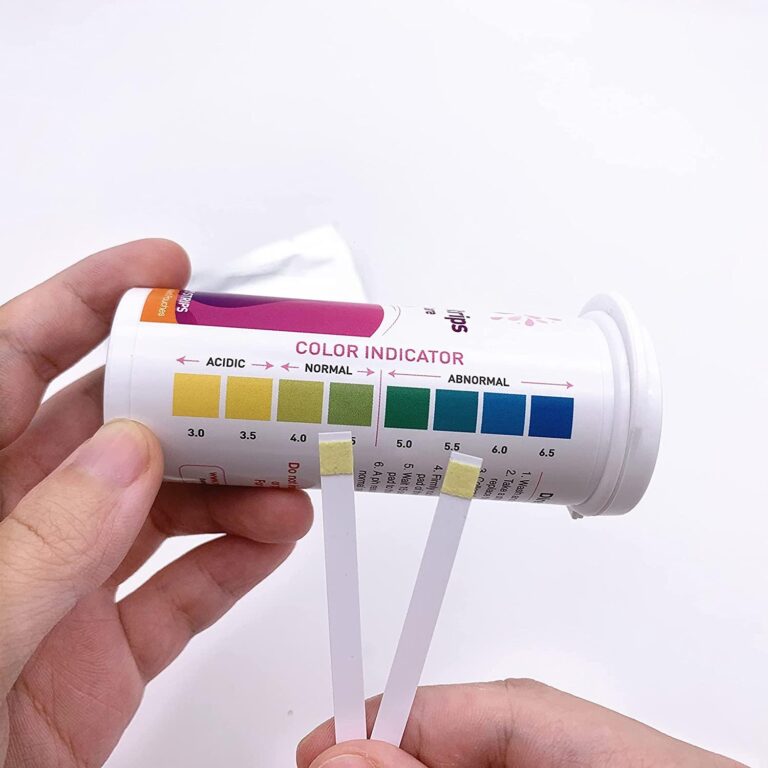
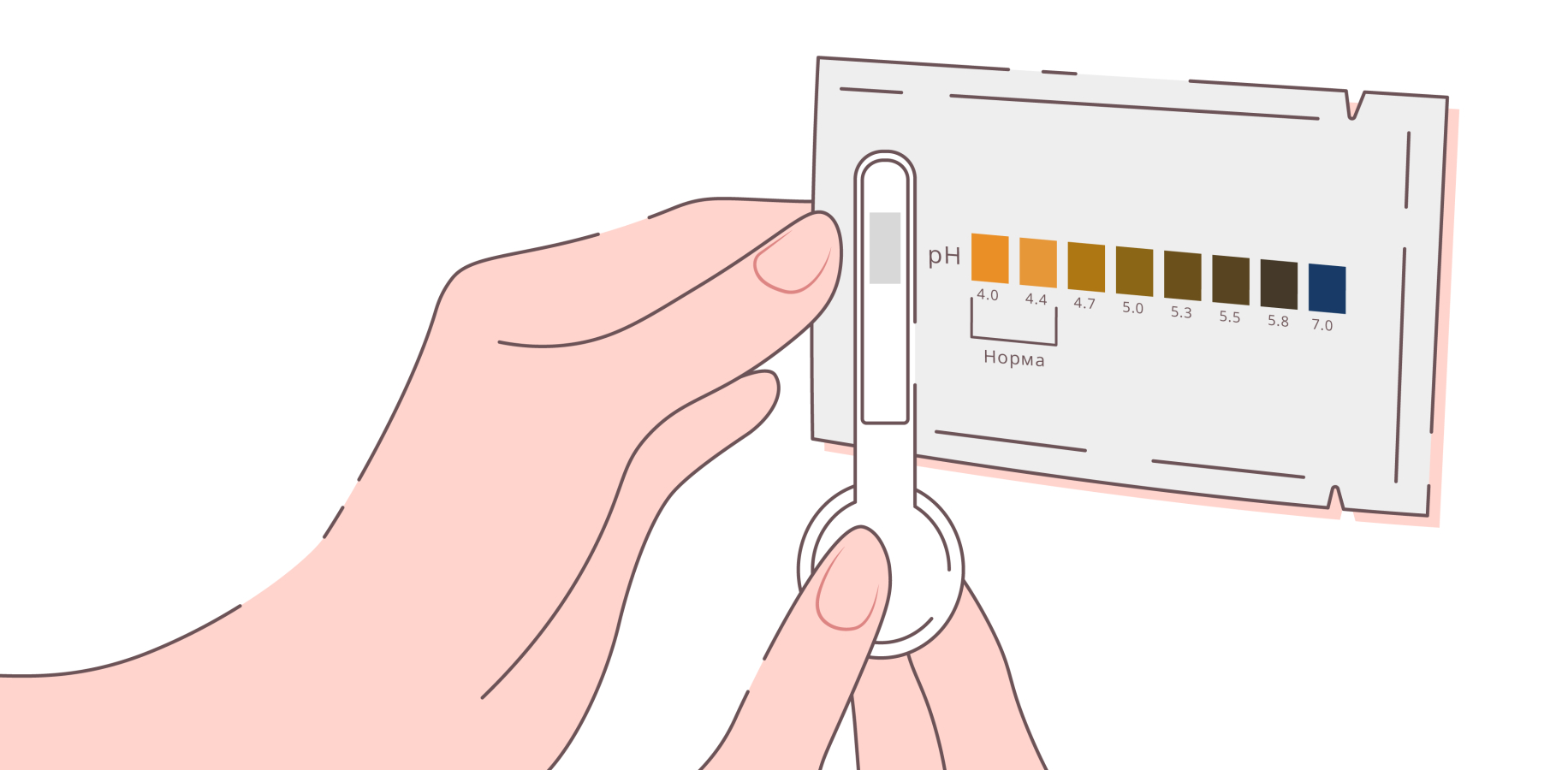
The mechanism: Not every episode of discomfort, discharge, or unusual smell is an active infection. Bacterial vaginosis and yeast infections present with overlapping symptoms yet have opposite treatment requirements. Antibiotics treat BV but can worsen or trigger a yeast infection. Using the wrong treatment, or using any treatment unnecessarily, disrupts the very pH balance you are trying to protect. At-home vaginal pH test strips allow you to measure your current pH level, typically within a range of 1 to 14, using a sample from vaginal discharge applied to a strip. A pH above 4.5 suggests possible bacterial vaginosis or trichomonas; a normal or low pH during unusual symptoms is more consistent with a yeast infection.
The evidence level: Clinical consensus holds that pH testing is a reliable first-line indicator when combined with symptom assessment. Research in reproductive medicine confirms that self-testing pH does not replace laboratory diagnosis but significantly improves the accuracy of self-guided decisions.
Practical implementation: Keep a pack of vaginal pH strips at home, available from most pharmacies without a prescription. If your reading comes back above 4.5 alongside odour or discharge changes, this warrants a GP or gynaecology appointment rather than self-treating. If your reading is 4.5 or below and your symptoms are itching-dominant with cottage-cheese-like discharge, a yeast infection is more probable. Tracking your pH readings across your menstrual cycle over two to three months will also help you identify your personal pH pattern and catch disruptions early.
Secret 2: Align Your Intimate Hygiene Routine with Your Menstrual Cycle
The mechanism: Your vaginal pH does not stay constant across the month. It shifts in a predictable hormonal rhythm. During the follicular phase, in the first half of your cycle, rising oestrogen supports robust Lactobacillus activity and keeps pH low. Just before ovulation and during menstruation, pH rises naturally, sometimes reaching 6.0 or higher, because menstrual blood itself has a pH of approximately 7.4. This temporary alkalinity is normal but creates a window of increased susceptibility to bacterial growth. Understanding this rhythm allows you to adapt your hygiene habits accordingly rather than using a one-size-fits-all daily routine.
The evidence level: Research suggests that symptomatic vaginal infections cluster around menstruation and in the immediate post-menstrual phase for a clear biological reason: pH disruption is at its peak. Clinical consensus in gynaecology holds that targeted hygiene adjustments during these windows can reduce recurrence of bacterial vaginosis by a clinically meaningful margin.
Practical implementation: During menstruation, rinse the external vulva with plain warm water only, more frequently than usual if you find this comfortable, to clear blood and reduce alkaline exposure time. Change menstrual products regularly. If you use a menstrual cup, ensure it is thoroughly cleaned between uses. In the days following your period, this is when the vaginal ecosystem is re-establishing itself. Avoid any new products, tight clothing, or sexual activity without a barrier method during this window, particularly if you have a history of recurrent BV.
In the mid-cycle phase, when oestrogen peaks and Lactobacillus activity is strongest, your vaginal pH balance is at its most resilient. This is a good time to note how your body feels without intervention, so you build an accurate baseline for comparison.
Secret 3: Harness the Protective Power of a Lactobacillus-Rich Diet
The mechanism: The Lactobacillus bacteria that maintain vaginal acidity are influenced not just by topical products or medications but by what you eat. A diet high in refined sugar feeds the proliferation of Candida albicans, the fungus responsible for yeast infections. Diets high in ultra-processed foods and low in fermented foods reduce the diversity and resilience of your overall microbiome, including the vaginal microbiome, because the gut and vaginal ecosystems are closely connected through shared bacterial populations and systemic immune signalling. Conversely, dietary choices that support a healthy gut Lactobacillus population appear to positively influence vaginal flora over time.
The evidence level: There is growing evidence from microbiome research that dietary fibre, fermented foods, and reduced sugar intake improve Lactobacillus dominance in both gut and vaginal environments. While the gut-vaginal microbiome axis is still an active area of research, clinical patterns consistently support its significance.
Practical implementation: Rather than overhauling your entire diet, focus on two specific changes. First, reduce added sugar. This does not mean eliminating fruit or complex carbohydrates. It means reducing foods where sugar is a primary ingredient: soft drinks, confectionery, sweetened yoghurts, and ultra-processed snacks. Second, introduce one to two portions of fermented food daily. Natural live-culture yoghurt, kefir, kimchi, sauerkraut, and miso all contain lactic acid bacteria. These are not necessarily the exact strains that colonise the vagina, but they support overall microbiome health in ways that downstream benefit vaginal flora.
Track whether changes in your diet correspond with changes in how frequently you experience infections over a two to three month period. The relationship is not immediate, but it is real and measurable.
Secret 4: Choose the Right Probiotic Strain for Vaginal pH Support
The mechanism: Not all probiotics are created equal. General gut probiotics, particularly those marketed broadly for digestive health, contain strains that do not typically colonise the vaginal tract. For vaginal pH support specifically, two Lactobacillus strains have the strongest clinical evidence: Lactobacillus rhamnosus GR-1 and Lactobacillus reuteri RC-14. When taken orally, these strains have been shown in randomised controlled trials to migrate from the gut to the vaginal environment via the ascending perineal route and to establish themselves in the vaginal microbiome, where they produce hydrogen peroxide and lactic acid, both of which lower vaginal pH and inhibit the growth of bacterial vaginosis-associated pathogens.
The evidence level: Research suggests that these two specific strains, when taken together, significantly reduce the recurrence of bacterial vaginosis when used alongside antibiotic treatment and can prevent BV episodes when used consistently in women with a history of recurrence. This is not the same as every probiotic supplement on the market making this claim.
Practical implementation: When selecting a probiotic, check the label for the specific strain names: L. rhamnosus GR-1 and L. reuteri RC-14. These are distinct from general “Lactobacillus rhamnosus” or “L. acidophilus” products. A daily oral dose taken consistently, ideally at the same time each day, is more effective than intermittent use. Vaginal probiotic suppositories containing these strains are also available and deliver the bacteria directly to the site of action, though oral supplementation has equivalent evidence in many studies. Always speak with your GP or gynaecologist before beginning supplementation if you are currently being treated for an active infection.
Healthline’s evidence-based guide to probiotics for vaginal health provides accessible background on how Lactobacillus strains function in clinical research contexts, which can help you ask better-informed questions at your next appointment.
Secret 5: Rethink Everything You’ve Been Told About Intimate Hygiene Products
The mechanism: The intimate hygiene market is large, lucrative, and, from a clinical standpoint, largely unnecessary and frequently harmful to vaginal pH balance. Vaginal douches, scented washes, deodorising sprays, scented panty liners, and perfumed soaps all share a common problem: they are alkaline or chemically complex in ways that disrupt the acidic vaginal environment. The vagina is self-cleaning. The discharge you produce is not a hygiene failure; it is an active biological process. Washing inside the vaginal canal removes the very Lactobacillus colonies your body has worked to establish. Even water introduced inside the vagina can temporarily raise pH and flush protective bacteria.
The evidence level: Clinical consensus is clear and consistent on this point: internal vaginal washing of any kind is not recommended. Studies examining women who douche regularly show significantly elevated rates of bacterial vaginosis, pelvic inflammatory disease, and disrupted vaginal microbiomes compared to women who do not. The external vulva, the skin around the vaginal opening, is a different matter and can be gently cleansed with a pH-balanced, unscented wash or warm water.
Practical implementation: Limit intimate cleansing to the external vulva only. Use warm water, or a fragrance-free, pH-balanced wash specifically formulated for external vulval use (with a pH between 3.5 and 4.5, which some products label explicitly). Avoid any product that contains: perfume or fragrance, antibacterial agents such as triclosan, glycerin in high concentrations, or parabens. After using the toilet, wipe front to back. After sexual activity, a gentle external rinse with warm water is sufficient. If you’re experiencing discharge that feels excessive or unusual, resist the urge to douche. That urge is understandable, but douching will worsen, not resolve, an underlying pH imbalance.
Secret 6: Understand How Sexual Activity Affects Your Vaginal pH Balance
The mechanism: Sexual activity is one of the most common triggers of vaginal pH disruption, and it is also one of the most frequently unreported topics in routine gynaecology consultations. Semen has a pH of approximately 7.1 to 8.0, making it significantly more alkaline than a healthy vagina. During unprotected penetrative sex, the introduction of semen temporarily raises vaginal pH. For most women, the vaginal microbiome recovers within a few hours. But in women with an already compromised or less resilient Lactobacillus population, this temporary alkaline shift can be enough to trigger a BV episode. This explains a pattern that many women notice but feel embarrassed to mention: recurring BV or unusual discharge in the days following sex with a regular partner.
The evidence level: Research suggests that semen-associated pH disruption is a recognised trigger for BV recurrence, particularly in women with low Lactobacillus diversity. Clinical consensus also holds that male partners can carry BV-associated bacteria on the penis without symptoms, creating an ongoing cycle of reinfection that antibiotics alone cannot break.
Practical implementation: Using condoms during penetrative sex is the single most effective mechanical method of protecting vaginal pH during sexual activity. This is not only about preventing sexually transmitted infections. It literally prevents pH-disrupting semen from contacting the vaginal environment. If you are in a long-term relationship and your BV keeps recurring despite antibiotic treatment, a conversation with your gynaecologist about concurrent male partner treatment may be warranted, though this is not yet standard protocol everywhere. After sex, a gentle external rinse is all that is needed. Avoid internal washing. If you use lubricants, check that they are pH-balanced and free from glycerin, which can feed Candida growth.
It is also worth noting that oral sex can introduce oral bacteria into the vaginal environment, and arousal itself involves natural fluid changes in vaginal pH. None of this means you should change your intimate life. It means understanding that sexual activity is a normal variable in vaginal pH management, not a source of shame.
Secret 7: Address the Stress-Hormone-pH Connection That Nobody Talks About
The mechanism: Chronic stress is one of the least-discussed but clinically significant disruptors of vaginal pH balance. Here is why. Prolonged psychological stress elevates cortisol, your body’s primary stress hormone. Elevated cortisol suppresses oestrogen production. Oestrogen is not merely a reproductive hormone. It has a direct structural role in the vaginal wall, maintaining the thickness and glycogen content of vaginal epithelial cells. Lactobacillus bacteria feed on that glycogen to produce lactic acid. Less oestrogen means less glycogen. Less glycogen means less Lactobacillus activity. Less Lactobacillus activity means rising pH. This chain reaction explains why many women notice more frequent infections during high-stress periods, around exam seasons, job changes, relationship difficulties, or bereavement. The connection is not imagined. It is hormonal and measurable.
The evidence level: Research suggests that psychosocial stress is an independent risk factor for bacterial vaginosis. Studies examining stress cortisol levels in women with recurrent BV consistently find elevated stress markers, even after controlling for other variables. This is a growing area of research in reproductive endocrinology, and it is beginning to inform clinical guidance in integrative gynaecology.
Practical implementation: Addressing stress as a component of vaginal health is not about achieving perfect inner peace. It is about identifying whether chronic stress is a pattern in your recurrence cycle. Keep a simple log alongside your pH readings: note major stressors and sleep quality alongside any symptoms or positive pH test results. Over several months, patterns frequently become visible.
From a clinical standpoint, interventions that reduce cortisol, including regular moderate exercise, improved sleep hygiene, mindfulness-based stress reduction, and, where appropriate, psychological support, have documented hormonal benefits that extend to oestrogen support and, by extension, vaginal pH balance. The vagina does not exist in isolation from the rest of your physiology. It responds to your whole body’s stress load.
Secret 8: Review the Medications That Quietly Disrupt Your Vaginal Microbiome
The mechanism: Several categories of commonly prescribed or commonly used medications have documented effects on vaginal pH balance and the vaginal microbiome. The most widely known is broad-spectrum antibiotics. When antibiotics are prescribed for a chest infection, urinary tract infection, or dental procedure, they eliminate bacteria without discrimination. Lactobacillus colonies are collateral damage. This is why antibiotic courses are frequently followed by yeast infections: the Candida fungi, which were previously kept in check by Lactobacillus acidity, suddenly find an undefended environment in which to multiply.
The combined oral contraceptive pill is a second significant but less-discussed disruptor. By suppressing oestrogen fluctuations, the pill can reduce the glycogen availability in vaginal epithelial cells that Lactobacillus depends upon. Some women notice recurring BV or a shift in their vaginal discharge pattern after starting hormonal contraception. This is a real biological mechanism, not a placebo effect.
Antihistamines, particularly first-generation formulations, reduce mucous membrane secretions throughout the body, including vaginal lubrication. This can raise vaginal pH by reducing the fluid medium in which Lactobacillus operates. Women who take antihistamines regularly for allergies may notice increased vaginal dryness and, in some cases, increased susceptibility to pH disruption.
The evidence level: Clinical consensus holds that antibiotic-associated yeast infections and vaginal microbiome disruption are well-documented. The impact of hormonal contraception on vaginal flora is an area of active clinical research, and the evidence, while still evolving, consistently supports an association between progestogen-dominant formulations and altered vaginal microbiome composition.
Practical implementation: If you are prescribed a course of antibiotics, begin an evidence-based vaginal probiotic (L. rhamnosus GR-1 and L. reuteri RC-14) simultaneously, and continue for at least four weeks after the course ends. Do not wait until symptoms develop. Speak with your prescribing doctor or pharmacist about taking probiotics during antibiotic therapy.
If you are on hormonal contraception and experiencing recurrent vaginal infections, it is worth discussing the formulation with your gynaecologist. Switching to a lower-dose combined pill, a progestogen-only option, or a non-hormonal method may significantly alter your pattern of infections. This is not a decision to make lightly or alone, but it is a clinically legitimate conversation to have.
Keep a medication history alongside your symptom diary. The picture this creates over three to six months is often more informative than any single appointment.
The Clinical Insight: What 19 Years in Practice Has Taught Me
In my 19 years of clinical practice, what I’ve seen most often is a woman who has been treated correctly for bacterial vaginosis four, five, sometimes eight times, given the right antibiotics, seen her symptoms clear, and then watched them return with dispiriting reliability, and who has never once been asked about her stress levels, her contraceptive method, her partner’s role in reinfection, or the state of her diet. She has been treated for an infection. She has not been treated as a whole person with an ecosystem.
The limitations of standard care here are not a reflection of individual clinicians failing their patients. They are a reflection of appointment times that rarely allow for systems-level thinking, and of a medical tradition that has historically treated the vagina as a symptomatic site rather than a dynamic, hormonally responsive environment.
What I have learnt from years of working with women experiencing recurrent intimate infections is this: the infection is almost never the whole story. It is the signal. The real story is about pH resilience. Some women have vaginal microbiomes dominated so thoroughly by Lactobacillus that extraordinary circumstances are required to displace them. Others have more variable microbiomes that are more sensitive to disruption. Neither is a moral failing. Both are manageable with the right information.
As I’ve seen with many patients, the turning point almost always comes when a woman stops treating individual episodes and starts understanding the underlying pattern. That shift, from reactive to proactive, from treating symptoms to supporting a system, is where lasting change begins.
When to See a Specialist: Red Flags You Should Not Wait On
Not every vaginal symptom can or should be managed at home. There are specific presentations that require prompt clinical assessment, and being specific about them matters more than vague reassurance.
If you experience a strong, fishy odour that does not resolve within five to seven days of your period ending, book an appointment with your GP or gynaecologist. This is a classic presentation of bacterial vaginosis that has not self-resolved, and it warrants laboratory confirmation before treatment.
If you experience vaginal itching, burning, or discharge that significantly interferes with your daily life or sleep for more than seventy-two hours, do not delay in seeking an assessment. This applies even if you think you already know what the cause is.
If you have experienced three or more episodes of bacterial vaginosis in the past twelve months, you meet the clinical threshold for recurrent BV and should be referred to a specialist gynaecologist rather than continuing to manage this with single-course antibiotic treatment. A specialist can offer extended suppressive antibiotic therapy, microbiome assessment, and partner treatment protocols that your GP may not have the capacity to coordinate.
If you experience vaginal dryness alongside recurring pH disruption, and you are over 40 or perimenopausal, request an assessment with your gynaecologist specifically for genitourinary syndrome of menopause (GSM), formerly known as vulvovaginal atrophy. This is a distinct hormonal condition that significantly reduces Lactobacillus activity and requires targeted treatment, often vaginal oestrogen, not standard anti-infective therapy.
If you notice any new lumps, sores, skin changes, or ulcerations on the vulva or vagina at any age, see a gynaecologist promptly. These are not typical symptoms of pH imbalance and require proper clinical evaluation.
Finally, if you are pregnant and experiencing any change in vaginal discharge or pH, consult your midwife or obstetrician without delay. BV in pregnancy carries specific risks to the pregnancy itself and requires prompt assessment.
You Know More Than You Did Yesterday, and That Matters
If you have reached this point in the article, something has shifted. You now understand that vaginal pH balance is not a niche topic for medical professionals. It is the central mechanism governing your intimate health, and it responds to practical, evidence-based choices that you can begin making today.
The single most important takeaway from everything you have read is this: your infections are not random, and they are not your fault. They are the predictable consequence of a disrupted ecosystem, and ecosystems can be restored.
Your first concrete next step is straightforward. Start a vaginal health log. Note your cycle day, any symptoms, any products you have used, your stress levels out of ten, and whether you used barrier contraception during sex. After eight to twelve weeks, patterns will emerge that will make your next clinical appointment far more productive.
You do not have to keep cycling through antibiotics and anxiety. You can understand the system, support it, and advocate for yourself within it.
Read next: [How to Talk to Your Gynaecologist About Recurrent BV Without Being Dismissed]
If this article helped you, share it with a friend who has been quietly struggling with the same questions. She deserves this information too.
Drop a comment below if you have questions or if there is a specific aspect of vaginal pH balance you would like explored in more depth. I read every one.
Medical Disclaimer
This article is for informational purposes only and does not constitute medical advice. Always consult a qualified healthcare provider before making any changes to your health or treatment plan.