Cortisol Imbalance: 6 Proven Effects on Weight Gain and Mental Health
The Moment You Stopped Recognising Your Own Body
It started gradually. Maybe you noticed it first in the mirror, a thickness around your waist that had not been there before. Or perhaps it was the anxiety that arrived without warning, cresting before you had even got out of bed. The sleep that no longer restored you. The fog that sat behind your eyes by mid-afternoon, making even simple conversations feel effortful.
You have had your blood work done. Your thyroid came back normal. Your iron levels were fine. Your GP said everything looked reasonable for your age. And yet here you are, feeling thoroughly unreasonable.
Here is what nobody checked. Here is what rarely makes it onto the standard panel despite being one of the most clinically significant hormonal drivers of weight and mood in women between thirty and fifty.
Cortisol imbalance.
Not a dramatic pathology. Not Cushing’s syndrome. Not adrenal failure. Something subtler, more widespread, and far more disruptive than its absence from standard care would suggest.
This article is for the woman who has been told she is fine but does not feel fine. It covers six of the most well-documented, clinically significant ways that cortisol imbalance reshapes your body and your mental health, and what you can actually do about each one.
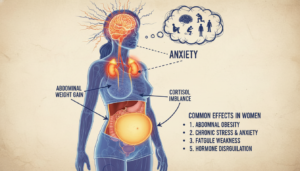
What Cortisol Imbalance Actually Is: The Clinical Foundation
Cortisol is produced by your adrenal glands, two small structures that sit atop each kidney. It belongs to a class of hormones called glucocorticoids, which are steroid hormones that regulate a wide range of physiological functions, including immune response, blood sugar control, metabolism, sleep architecture, and the stress response itself.
Under normal conditions, cortisol follows a reliable daily rhythm. Levels are highest within thirty minutes of waking, a phenomenon known as the cortisol awakening response, and they decline steadily through the day, reaching their lowest point between midnight and approximately four in the morning. This rhythm is coordinated by the hypothalamic-pituitary-adrenal axis, commonly called the HPA axis, which is the hormonal communication network between your brain and your adrenal glands.
Think of the HPA axis like a sophisticated thermostat. When the temperature drops, the system activates. When it rises, the system reduces output. The problem arises when the thermostat gets stuck. Chronic stress, poor sleep, blood sugar instability, inflammation, and accumulated trauma can all dysregulate this feedback loop, leaving cortisol persistently elevated, measurably low at the wrong times, or dysrhythmic in a pattern that disrupts every system it touches.
Cortisol imbalance refers to any sustained disruption of this daily cortisol rhythm, encompassing chronic elevation, blunting, or dysrhythmia. It is not a single abnormal test result. It is a pattern.
The reason this topic is so underserved in mainstream medicine is straightforward. Cortisol is only routinely tested when a clinician suspects a specific pathological condition, and the reference ranges used for standard blood cortisol tests reflect disease states rather than functional optimal ranges. The vast middle ground, where cortisol is technically within range but functionally disrupted, remains largely invisible to routine care.
The six effects that follow are each supported by clinical research and are commonly seen in women navigating this middle ground.
The 6 Proven Effects of Cortisol Imbalance on Weight Gain and Mental Health
Effect 1: Preferential Accumulation of Visceral Abdominal Fat
Why Cortisol Imbalance Targets Your Middle
If there is one physical manifestation of cortisol dysregulation that women report most consistently, it is this: weight gathering specifically around the abdomen, even when diet and exercise have not changed. This is not coincidence. It is anatomy.
Visceral fat, which is the fat stored deep within the abdominal cavity around your organs, contains a significantly higher concentration of glucocorticoid receptors than subcutaneous fat, which is the fat directly beneath your skin. Glucocorticoid receptors are the docking sites for cortisol on the surface of fat cells. When cortisol is chronically elevated, these receptors are repeatedly activated, sending a sustained biochemical signal to deposit fat specifically in the abdominal region.
There is a second, compounding mechanism. Cortisol actively opposes the action of insulin, the hormone responsible for moving glucose from your bloodstream into your cells. When cortisol is chronically high, insulin resistance develops gradually. Your body produces more insulin to compensate for its reduced effectiveness, and persistently elevated insulin is, physiologically speaking, a fat-storage signal. The result is a dual driver of abdominal fat accumulation: direct cortisol-mediated deposition and insulin-driven energy storage, operating simultaneously.
The Cruel Irony of Caloric Restriction
What makes this particularly frustrating is that the most intuitive response, eating less, frequently makes the situation worse. Severe caloric restriction is itself a physiological stressor. It activates the HPA axis. It raises cortisol. This is why women in a state of cortisol dysregulation often find that their attempts to diet more strictly are met with paradoxical weight retention or continued abdominal expansion, regardless of their caloric deficit.
Clinical consensus holds that cortisol-driven abdominal fat accumulation requires addressing the cortisol pattern directly, not simply reducing energy intake. Approaches that stabilise blood sugar, improve sleep quality, and reduce physiological stress load are more likely to shift this type of weight than restriction alone.
The Metabolic Consequences That Compound Over Time
Visceral fat is not metabolically inert. It is an active endocrine tissue that secretes inflammatory signalling proteins called cytokines. These cytokines maintain a state of low-grade systemic inflammation that further stresses the HPA axis, prompting additional cortisol release. The loop tightens. The abdominal fat drives more cortisol, which drives more abdominal fat. Understanding this mechanism should meaningfully change how you approach weight management if cortisol dysregulation is part of your picture.
Effect 2: Relentless Carbohydrate Cravings and Compulsive Eating Patterns
The Biochemical Drive Beneath the Craving
You reach for bread at four in the afternoon. You find yourself finishing the biscuits you had no intention of touching. You eat a reasonably balanced dinner and then, an hour later, feel an insistent pull towards something sweet. You tell yourself this is a lack of discipline. It is not. It is a cortisol-mediated neurobiological cascade.
Here is the mechanism. Cortisol’s evolutionary role is to mobilise energy in the face of perceived threat. It does this primarily by stimulating the liver to release stored glucose into the bloodstream, a process called gluconeogenesis. Under chronic stress, this glucose release occurs repeatedly but without the corresponding physical activity that would normally burn it. Blood sugar rises. Insulin is secreted to manage the rise. Blood sugar then falls below its starting point. This state, called reactive hypoglycaemia, which is a temporary drop in blood sugar following an insulin spike, activates your brain’s emergency hunger signal with genuine urgency.
Your brain does not register this as a lifestyle choice. It registers it as an energy crisis. And the fastest resolution to an energy crisis is rapidly digestible carbohydrate. The craving is not a character flaw. It is a survival mechanism responding to a hormonal environment you did not choose.
Serotonin, Tryptophan, and the Carbohydrate Connection
There is an additional layer to this that explains why the cravings cluster so reliably in the late afternoon and evening. Carbohydrates, particularly starchy and sweet foods, stimulate insulin release. Insulin facilitates the transport of tryptophan, an amino acid, across the blood-brain barrier. Tryptophan is the direct precursor to serotonin, your primary mood-regulating neurotransmitter. Chronically elevated cortisol suppresses serotonin synthesis and receptor sensitivity.
Women experiencing cortisol dysregulation are frequently operating with a genuine serotonin deficit. When they reach for carbohydrates in the evening, their brains are attempting to self-medicate this deficit. The relief is real, brief, and biological. The blood sugar crash that follows an hour later restarts the cycle.
Understanding this does not eliminate the craving. But it should fundamentally change how you speak to yourself about it.
The Long-Term Metabolic Risk
Repeated blood glucose spikes and corresponding insulin surges, sustained over months and years, progressively erode insulin sensitivity. This trajectory, from cortisol dysregulation to blood sugar instability to insulin resistance, is associated in the research literature with an elevated risk of developing type 2 diabetes and polycystic ovary syndrome, a condition that itself involves measurable HPA axis dysfunction in many women.
Research suggests that interventions targeting cortisol normalisation, alongside blood sugar stabilisation through regular, balanced eating, can meaningfully improve insulin sensitivity before any weight change occurs. This is clinically important because it means the metabolic benefit of addressing cortisol precedes and does not depend on weight loss.
Effect 3: Persistent Low Mood, Generalised Anxiety, and Emotional Dysregulation
How Cortisol Physically Alters Your Brain
The relationship between cortisol imbalance and mental health is not psychological in origin. It is structural and neurochemical. Chronically elevated cortisol causes measurable, documentable changes to brain architecture that directly produce the mood symptoms women in this situation describe.
The hippocampus, a paired brain structure embedded in the temporal lobe and central to emotional memory, stress contextualisation, and the regulation of the fear response, is among the most cortisol-sensitive regions in the brain. It contains an exceptionally high density of glucocorticoid receptors. Under chronic cortisol exposure, research has consistently shown suppression of neurogenesis in the hippocampus, which is the production of new neurons, alongside, in more severe and prolonged cases, a measurable reduction in hippocampal volume.
A hippocampus functioning below its optimal capacity is less able to contextualise incoming stressors, less able to signal the HPA axis that a perceived threat has passed, and less effective at regulating the emotional intensity of the amygdala, which is the brain’s threat-detection centre. The practical consequence is that stressors which would previously have felt manageable begin to feel genuinely overwhelming. The emotional volume is turned up, and your brain’s mechanism for turning it back down is compromised.
The Neurotransmitter Deficit at the Root of Anxiety and Depression
Chronic cortisol elevation simultaneously disrupts the production and receptor sensitivity of multiple neurotransmitters involved in mood regulation. Serotonin, as discussed, is suppressed. Dopamine receptor sensitivity is blunted, reducing motivation and the capacity for pleasure. GABA, which is your brain’s primary inhibitory neurotransmitter and the neurochemical most directly responsible for the felt experience of calm and safety, is also suppressed under chronic cortisol exposure.
The resulting neurochemical environment closely resembles the substrate of clinical anxiety disorder and major depressive disorder. Women in this state are frequently treated for the symptom, with antidepressants, anxiolytics, or both. These interventions can provide meaningful and genuine relief for many. What they rarely do is address the cortisol dysregulation that may be contributing to, or in some cases primarily driving, the symptom picture.
This is not a criticism of psychiatric treatment. It is an argument for investigating cortisol as part of the clinical picture for women presenting with anxiety or depression, particularly when those symptoms coexist with the physical signs of cortisol imbalance.
The Perimenopause Complication
For women in their early to mid-forties, this neurochemical picture is further complicated by the hormonal shifts of perimenopause. Declining oestrogen is independently associated with increased HPA axis reactivity. As oestrogen levels fall, the cortisol response to psychosocial stressors tends to amplify. The symptoms of each condition reinforce those of the other. Women in this phase frequently present with worsening anxiety, new-onset low mood, sleep disruption, and weight changes that their clinicians may attribute entirely to perimenopausal hormonal transition without evaluating the cortisol component.
A thorough hormonal evaluation for women in this demographic should include both ovarian hormone assessment and HPA axis function. Treating them as separate investigations frequently leads to an incomplete picture.
Effect 4: Sleep Architecture Disruption and the Exhaustion-Cortisol Cycle
The Physiological Paradox of Being Tired and Wired
Sleep disruption is perhaps the most universally experienced symptom of cortisol imbalance, and it presents in a specific and recognisable pattern. You are profoundly tired by early evening. You fall asleep relatively easily. And then, somewhere between two and four in the morning, you are wide awake, your mind cycling through concerns and your body feeling oddly alert. Returning to sleep is difficult. By morning, you are as fatigued as when you went to bed.
This is not insomnia in the conventional sense. It is cortisol dysrhythmia, specifically a pattern in which cortisol fails to suppress adequately in the late-night period when it should be at its nadir.
Cortisol and melatonin exist in an inverse relationship. As cortisol falls in the evening, melatonin, the hormone that signals sleep readiness, rises. When cortisol remains elevated or rises inappropriately in the late-night hours, melatonin production is suppressed or delayed. The sleep-onset signal either arrives late or is insufficient to maintain the deep, slow-wave sleep phases that are physiologically restorative.
The Specific Damage to Slow-Wave and REM Sleep
Cortisol does not merely delay sleep onset. Research suggests it preferentially disrupts the most restorative stages of sleep. Slow-wave sleep, also called deep sleep or stage three NREM sleep, is the phase during which the body undertakes physical repair, growth hormone secretion occurs, and the immune system is most active. REM sleep is critical for emotional processing, memory consolidation, and cognitive integration.
When nocturnal cortisol elevation reduces the proportion of these stages in your sleep architecture, you wake feeling unrefreshed even after seven or eight hours in bed. Physical recovery is impaired. Emotional regulation the following day is compromised. Cognitive performance is reduced. And the sleep debt accumulated over weeks and months becomes a physical burden in its own right.
Why Poor Sleep Makes Everything Worse
The particularly compounding dimension of this cycle is what sleep deprivation does to cortisol the following day. Even a single night of fragmented or insufficient sleep measurably elevates cortisol the next morning. Multiple disrupted nights consecutively push baseline cortisol higher and sustain elevated levels throughout the day. This means that poor sleep driven by cortisol dysregulation directly worsens the cortisol dysregulation causing it.
Clinical consensus holds that sleep restoration is one of the highest-leverage and most time-critical interventions in cortisol management. Before other strategies reach their full effectiveness, sleep must be addressed. This does not mean spending more hours in bed. It means creating the physiological conditions, through consistent sleep timing, evening light reduction, and nervous system downregulation practices, that allow cortisol to fall appropriately in the evening and melatonin to rise uninhibited.
Effect 5: Thyroid Suppression and the Hidden Metabolic Slowdown
The Cortisol-Thyroid Relationship Your Test Results Miss
This is perhaps the most clinically underappreciated effect on this list, and it explains a pattern that enormous numbers of women recognise but cannot get their doctors to take seriously: every symptom of an underactive thyroid, but a TSH result that comes back normal.
Your thyroid gland produces primarily thyroxine, known as T4, which is an inactive precursor hormone. T4 must be converted into its active form, triiodothyronine, known as T3, before it can enter cells and regulate metabolic rate. This conversion happens largely in peripheral tissues, including the liver, kidneys, and gut, and is dependent on specific enzymes.
Chronically elevated cortisol directly inhibits the enzyme responsible for this conversion. The result is a state of relative thyroid underactivity at the cellular level, even when thyroid hormone production from the gland itself is normal. This is sometimes referred to as low T3 syndrome or functional hypothyroidism. TSH, the pituitary signal that tells the thyroid to produce more hormone, may remain completely normal because the problem is not with thyroid production. It is with peripheral conversion.
What This Looks Like in Practice
Women in this state present with the classic symptom cluster of hypothyroidism: fatigue that is not resolved by sleep, hair thinning, unexplained weight gain, constipation, dry skin, cold intolerance, and a cognitive fog that makes concentration effortful. They are frequently told, after TSH testing, that their thyroid is fine. And technically, structurally, it is. The problem is downstream, at the level of hormone activation, and it is driven by cortisol.
A comprehensive thyroid evaluation that includes Free T3, Free T4, and reverse T3, alongside a cortisol assessment, offers a substantially more informative picture. As documented in Healthline’s overview of thyroid function and hormonal health, Free T3 is the most metabolically active form of thyroid hormone and its measurement is increasingly considered essential for women with thyroid-type symptoms and normal TSH results.
Requesting this panel specifically, rather than accepting TSH alone as a complete thyroid assessment, is a concrete step you can take in your next clinical appointment.
The Metabolic Consequence for Weight Management
When cellular thyroid activity is reduced, your basal metabolic rate, which is the number of calories your body burns at rest, falls. Your cells are less efficient at converting food into usable energy. Fatigue compounds because your muscles and organs are receiving less metabolic fuel than they require. Weight management becomes measurably harder through no fault of personal behaviour.
The further difficulty is that continued caloric restriction in this state signals the body to downregulate metabolic rate further. Cortisol accelerates this protective response. The result is a situation in which the obvious approach, eating less, deepens the metabolic slowdown it was intended to reverse.
Addressing cortisol dysregulation in this context frequently produces a secondary improvement in thyroid hormone conversion, with a gradual restoration of metabolic rate over the following weeks to months.
Effect 6: Cognitive Decline, Brain Fog, and the Loss of Mental Sharpness
When Your Mind Becomes Unfamiliar
Many women describe the cognitive effects of cortisol imbalance before they describe the physical ones. They use language like “thinking through treacle,” “losing words mid-sentence,” “walking into rooms and forgetting why,” and “feeling like a dimmer version of myself.” They frequently attribute these changes to ageing, menopause, or overwork, and they rarely mention them to a clinician because they feel too vague to be taken seriously.
They are not vague. They are measurable, physiologically grounded, and very specific to what chronic cortisol exposure does to the brain.
The hippocampus, already discussed in the context of emotional regulation, is equally central to memory formation and retrieval. Chronic cortisol suppression of hippocampal neurogenesis impairs both the encoding of new memories and the recall of existing ones. Women experiencing cortisol dysregulation frequently report forgetting recent conversations, repeating questions they have already asked, and losing confidence in their own memory in a way that is genuinely frightening.
Prefrontal Cortex Suppression and the Loss of Executive Function
Cortisol also directly suppresses activity in the prefrontal cortex, which is the region of your brain responsible for executive function, encompassing planning, prioritising, problem-solving, impulse control, and decision-making. Acute cortisol elevation temporarily suppresses this region to allow faster, more instinctive amygdala-driven responses, which is adaptive in a genuine emergency. Under chronic cortisol elevation, this suppression becomes semi-permanent.
The practical experience is one of reduced cognitive efficiency. Tasks that previously required no conscious effort now require deliberate energy. Decision-making feels laboured. The ability to hold multiple pieces of information in working memory simultaneously, to plan ahead, to follow complex conversations, and to filter relevant from irrelevant information is all reduced.
This is not a sign that you are losing your mind. It is a cortisol effect. And as the research literature suggests, it is substantially reversible with effective cortisol management.
Distinguishing Cortisol-Driven Brain Fog from Perimenopausal Cognitive Change
For women in their late thirties and forties, this cognitive picture overlaps significantly with the cognitive symptoms of perimenopause, which also include memory lapses, reduced concentration, and mental fatigue. The distinction matters because the interventions differ. Perimenopausal cognitive symptoms are driven largely by declining oestrogen and its effects on brain energy metabolism and acetylcholine function. Cortisol-driven cognitive symptoms are driven by HPA axis dysregulation and can worsen independently of oestrogen levels.
In many women, both processes are occurring simultaneously, compounding each other. An evaluation that assesses oestrogen alongside cortisol, conducted by a reproductive endocrinologist or a gynaecologist with expertise in hormonal health, is the appropriate approach for women presenting with cognitive symptoms in this age bracket.
The hopeful message here is this: the brain retains significant neuroplasticity at every age and stage. Cortisol-related cognitive changes, when addressed through effective cortisol regulation, show meaningful recovery in the research literature. Your brain’s capacity to restore clarity is greater than the symptom experience suggests.
How These Six Effects Create a Self-Sustaining Loop
Understanding each effect individually is useful. Understanding how they connect to each other is what transforms the picture from a bewildering list of unrelated symptoms into a coherent, addressable clinical reality.
Chronic stress elevates cortisol. Elevated cortisol disrupts sleep architecture. Disrupted sleep raises cortisol the next day. Elevated cortisol drives blood sugar instability, generating carbohydrate cravings and increasing abdominal fat deposition. Abdominal fat secretes inflammatory cytokines that further activate the HPA axis. Elevated cortisol suppresses thyroid conversion, slowing metabolic rate and deepening fatigue. Fatigue reduces capacity for physical activity, removing a key cortisol-regulatory mechanism. Cortisol-mediated changes to the hippocampus and amygdala amplify anxiety and worsen mood. Declining mood and worsening anxiety generate their own psychological stress load. That psychological stress raises cortisol.
This is not a linear chain with a clear beginning and end. It is a loop, and it can be entered and interrupted at multiple points. The question is not how to solve everything simultaneously. The question is which entry point is most accessible to you right now.
Evidence-Based Approaches to Supporting Cortisol Regulation
The following strategies are evidence-supported as meaningful interventions for cortisol dysregulation. They are not replacements for clinical investigation or treatment. They are what the research and clinical practice suggest are effective in parallel with professional guidance.
Prioritising Sleep Consistency Over Sleep Duration
The most important single variable in cortisol regulation is not how many hours you sleep, though duration matters. It is the consistency of your sleep and wake times. Research suggests that irregular sleep timing, varying your wake time by more than an hour between weekdays and weekends, is independently associated with elevated daytime cortisol and HPA axis dysregulation, even when total sleep duration appears adequate.
A consistent wake time, maintained even after a poor night, is the single most powerful anchor for the cortisol diurnal rhythm. It signals to the HPA axis when morning cortisol should peak, which in turn regulates the entire day’s cortisol curve.
Balanced, Regular Eating to Stabilise Blood Sugar
Rather than restricting calories, which activates the cortisol stress response, the evidence supports a pattern of regular, balanced eating that prevents the blood glucose drops that trigger cortisol-driven cravings. Including a source of protein, complex carbohydrate, and healthy fat at each meal slows glucose absorption and provides a steadier fuel supply, reducing the frequency and intensity of the reactive hypoglycaemia that drives compulsive eating.
Eating at regular intervals, approximately every three to four hours, is in itself a cortisol management strategy because it prevents the glucose scarcity signal that activates the HPA axis.
Movement That Supports Rather Than Further Stresses the System
Exercise is a well-established cortisol regulation tool, but the type and intensity matter considerably when cortisol is already elevated. High-intensity exercise, particularly training that involves extended periods of cardiovascular exertion, can produce acute cortisol spikes that worsen the overall picture in women already in a state of HPA axis strain.
Clinical consensus favours lower to moderate intensity movement during the cortisol recovery phase: walking, swimming, yoga, and gentle cycling are consistently supported by the research as effective cortisol-lowering modalities. As the system stabilises, higher-intensity exercise becomes more tolerable and beneficial again.
Physiological Downregulation of the Nervous System
The HPA axis does not distinguish between physical and psychological threat. Practices that actively shift the nervous system from sympathetic dominance, which is the activated, alert state that sustains cortisol output, to parasympathetic activity have documented physiological effects on cortisol levels.
Diaphragmatic breathing with an extended exhalation, specifically a breath pattern involving a four-second inhale and a six to eight second exhale, activates the vagus nerve and measurably reduces cortisol output in the short and medium term. According to Mayo Clinic’s guidance on stress management and its physiological effects, regular practices including mindfulness-based stress reduction, progressive muscle relaxation, and therapeutic social connection all produce quantifiable reductions in cortisol activity and are now considered evidence-based interventions rather than complementary wellness extras.
These are not soft recommendations. These are physiological interventions with documented mechanisms.
Reducing the Internal Stress of Self-Blame
This one deserves naming explicitly, because it is both clinically relevant and almost never mentioned. Many women with cortisol dysregulation carry enormous psychological weight around the physical changes they are experiencing. The unexplained weight gain generates shame. The cognitive fog generates fear. The emotional lability generates self-criticism. The eating patterns they describe are accompanied by intense guilt.
Shame, self-blame, and fear are each independently cortisol-activating states. The internal narrative of “I am failing my body” sustains the very hormonal environment it is responding to. This is not a criticism. It is physiology. And addressing it, ideally through therapeutic support, is itself a measurable cortisol management strategy.
The Clinical Insight: What I See That Standard Care Misses
In my 19 years of clinical practice, what I’ve seen most often is not the dramatic presentation. It is the woman who has been carrying this picture for three, four, five years and who has been told, repeatedly, that her results are normal. She has learnt to distrust her own perception of her body because the medical system has repeatedly told her that perception is inaccurate.
By the time she reaches my office, she is not just dealing with cortisol dysregulation. She is dealing with the accumulated psychological cost of having her experience dismissed. She is often apologetic about “over-reacting.” She qualifies her symptoms before she has even finished describing them.
The gap in standard care is not merely the absence of cortisol testing. It is the absence of clinical curiosity about the full hormonal picture in women presenting with this constellation of symptoms. TSH testing does not assess T3 conversion. Standard blood cortisol does not capture cortisol rhythm. Routine mood screening does not explore neurochemical drivers. Women receive piecemeal investigations and piecemeal diagnoses when what they need is a clinician looking at the whole interconnected system.
The single most important shift I can offer is this: if your results are normal but your experience is not, you have not reached the end of the investigation. You have reached the limit of what was tested.
When to See a Specialist: Specific Red Flags and Timeframes
The following presentations warrant specialist evaluation rather than watchful waiting. Each one specifies the symptom, the timeframe, and the clinician best positioned to investigate it.
If your waist circumference has increased by five centimetres or more over a period of twelve months, without a meaningful change in diet or physical activity level, and the weight appears concentrated specifically in the abdominal region, book an appointment with an endocrinologist. Request a four-point salivary cortisol test to assess your cortisol diurnal rhythm across the day, alongside fasting insulin and a full lipid panel.
If you are waking between two and four in the morning with alertness, a sense of dread, or a racing mind, and this has occurred more than three nights per week for four consecutive weeks, request a salivary cortisol rhythm test from your GP. A single morning blood cortisol provides limited information about rhythm and is insufficient as a standalone assessment.
If low mood or anxiety has persisted for longer than eight weeks and has not responded meaningfully to lifestyle changes or conventional treatment, speak to a reproductive endocrinologist or an integrative gynaecologist about requesting a hormonal panel that includes serum cortisol, Free T3, Free T4, DHEA-S (a companion adrenal hormone), and sex hormones including oestradiol and progesterone. These do not need to be investigated separately.
If you have thyroid-type symptoms, including persistent fatigue, hair thinning, constipation, and cold intolerance, with a TSH result within the normal reference range, specifically request Free T3 and reverse T3 alongside your TSH at your next GP appointment. If your GP declines, a private endocrinology consultation is a reasonable next step.
If cognitive symptoms, including significant memory lapses, word-finding difficulties, or concentration impairment, have worsened over a three to six-month period and are affecting your daily functioning, see a reproductive endocrinologist or a neurologist for a combined hormonal and cognitive assessment before attributing the change to age or stress alone.
Do not accept “everything is within normal limits” as a final answer when your experience does not reflect that. Normal reference ranges reflect population statistics, not individual optimal function.
You Are Not Failing. You Are Under-Investigated.
If reading this has produced the particular relief of recognition, the feeling that someone has finally described what has been happening in your body in terms that make structural sense, then hold onto that. That recognition matters.
Cortisol imbalance is not a character flaw. It is not the consequence of insufficient willpower, inadequate effort, or poor self-management. It is a physiological pattern with measurable causes and measurable solutions, and it deserves the same clinical rigour as any other hormonal condition.
The single most important takeaway from everything covered here is this: the symptoms you are experiencing have a physiological basis. They are not imagined, they are not exaggerated, and they are not simply the cost of being a woman of a certain age in a demanding world.
Your most concrete next step is to request a cortisol diurnal rhythm assessment, either through your GP or a private endocrinologist, alongside a full thyroid panel including Free T3. Take this article with you if it helps you articulate the clinical picture.
Read Next: “How to Prepare for a Hormonal Health Consultation: The Questions That Get Results.”
And if this has spoken to your experience, drop a comment below. Your words may be exactly what another woman needs to read tonight.
This article is for informational purposes only and does not constitute medical advice. Always consult a qualified healthcare provider before making any changes to your health or treatment plan.

