Stage 4 Breast Cancer Life Expectancy 2026: 8 Hopeful Advancements That Are Helping Women Live Longer Than Ever Before
By Dr. Naomi | Women’s Health Physician | webzalo.com
It Is 2:47am and You Cannot Stop Searching
You have typed it into Google again. “Stage 4 breast cancer life expectancy.” The results load. Five-year survival rate. Median survival time. Statistical averages presented in cold, clinical rows.
Maybe the diagnosis arrived last month. Maybe it came two years ago and there has just been a new development, a scan that changed things. Maybe it is not you at all. Maybe it is your mother, your sister, your closest friend, and you are sitting in the dark trying to understand what she is now facing.
Before you read anything else, here is what I need you to know.
The numbers you are finding online are already out of date.
The landscape of stage 4 breast cancer treatment has shifted so dramatically over the last five years that many of the survival figures circulating on the internet simply do not reflect what is clinically possible in 2026. Women are living longer. Women are living better. Some are achieving outcomes that would have seemed genuinely unthinkable a decade ago.
The research is real. The treatments are approved. The evidence is building fast.
This article is for you, wherever you are sitting tonight.
What Stage 4 Breast Cancer Actually Means, and Why the Old Survival Statistics No Longer Tell the Whole Story
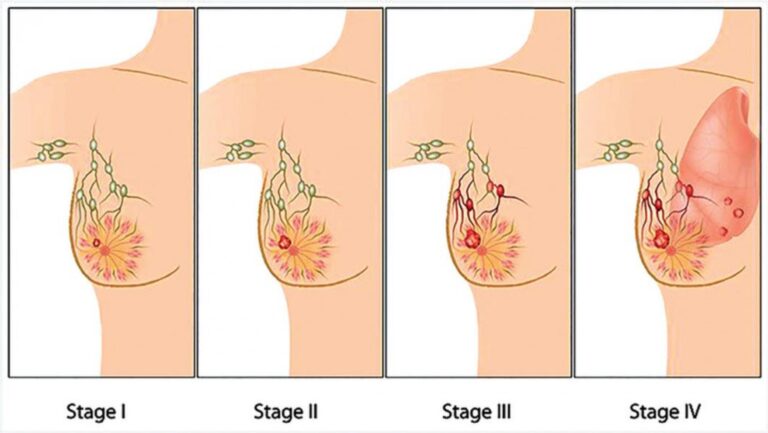
Stage 4 breast cancer, also known as metastatic breast cancer (MBC), is defined as breast cancer that has spread beyond the breast tissue and nearby lymph nodes to distant organs or tissues. The most common sites of spread include the bones, lungs, liver, and brain. The word “metastatic” simply means the cancer has travelled, or metastasised, to a location far from its point of origin.
Think of a stage 4 diagnosis less like a cliff edge and more like a fork in a long road. The path forward depends on where the cancer has spread, which molecular markers the tumour carries, which subtype of breast cancer it is, and what therapies your oncology team can deploy.
Here is the direct answer to the question most women ask first: Stage 4 breast cancer life expectancy in 2026 is no longer defined by a single statistic. According to data compiled by the National Cancer Institute’s SEER programme, the five-year relative survival rate for distant-stage breast cancer currently sits at approximately 28 to 30 percent. However, this figure draws on data from patients diagnosed in earlier years, before the most transformative treatments of today became available. Women diagnosed and treated right now, with access to current therapies, are achieving outcomes that significantly exceed these historical figures.
This distinction matters enormously. Survival statistics always lag behind clinical reality by several years. A five-year survival figure published today reflects the outcomes of women treated five to ten years ago, using tools that have often already been superseded.
One reason this topic is so profoundly underserved in mainstream health communication is precisely this lag. New treatments enter clinical use. Trials read out. Approvals are granted. But the population-level statistics that most people encounter online take years to catch up. By the time a headline survival figure reflects a new drug, that drug may already have been surpassed by the next generation.
What you need, then, is not just a statistic. You need to understand what is changing right now, at the level of actual clinical medicine. That is what this article is here to give you.
Understanding Your Subtype: Why Not All Stage 4 Breast Cancer Is the Same
Before examining the advancements themselves, it is worth briefly clarifying that metastatic breast cancer is not a single disease. Its molecular subtypes behave differently and respond to entirely different classes of treatment. This distinction is clinically critical.
Hormone receptor-positive, HER2-negative (HR+/HER2-) is the most common subtype, accounting for roughly 70 percent of all breast cancers. These tumours are driven by oestrogen or progesterone, making them responsive to hormone-blocking therapies. Most of the CDK4/6 inhibitor and endocrine therapy advances described below apply to this group.
HER2-positive breast cancer accounts for approximately 15 to 20 percent of cases. These tumours overexpress a protein called HER2, which stands for human epidermal growth factor receptor 2, a driver of rapid tumour cell division. Targeted anti-HER2 therapies have transformed outcomes for this subtype more than almost any other treatment development in the last two decades.
Triple-negative breast cancer (TNBC) represents around 10 to 15 percent of cases. These tumours lack oestrogen receptors, progesterone receptors, and HER2 expression. Historically the hardest to treat, TNBC has seen meaningful progress through immunotherapy combinations.
Knowing exactly which subtype you are dealing with is not just useful contextual information. It is the clinical foundation on which all treatment decisions in metastatic breast cancer should be built. If you are unsure of your receptor status, ask your oncologist to clarify this at your next appointment. It will help you make sense of every advancement described below.
8 Hopeful Advancements in Stage 4 Breast Cancer Life Expectancy 2026
These are not hypothetical possibilities or distant research promises. These are real, approved or rapidly emerging treatments that are reshaping survival outcomes for women with metastatic breast cancer right now.
1. CDK4/6 Inhibitors: Putting the Brakes on Tumour Cell Division
For women with HR+/HER2- metastatic breast cancer, CDK4/6 inhibitors have arguably been the single most transformative class of drugs in the past decade. Understanding why they work requires a brief look at the cell cycle.
CDK4 and CDK6 are proteins called cyclin-dependent kinases. In healthy tissue, they act as gatekeepers, regulating the precise moment when a cell is permitted to divide and replicate. In hormone receptor-positive breast cancer, this gating mechanism is frequently overridden, and cells divide in an uncontrolled, rapid cascade. CDK4/6 inhibitors work by blocking these proteins, essentially reinstating the brakes on tumour cell division.
Used in combination with standard hormone-blocking therapy, typically an aromatase inhibitor or fulvestrant, CDK4/6 inhibitors have produced results in clinical trials that represent a genuine step change in outcomes.
Data from multiple large-scale randomised controlled trials have demonstrated that adding a CDK4/6 inhibitor to hormone therapy can more than double progression-free survival compared to hormone therapy alone. Crucially, follow-up data from several of these trials have also confirmed an improvement in overall survival. This is an important distinction in oncology research. Demonstrating that a drug helps women live longer without disease progression is meaningful. Demonstrating that it helps women simply live longer is the gold standard, and CDK4/6 inhibitors have cleared that bar.
Three CDK4/6 inhibitors are currently in widespread clinical use internationally. They differ slightly in their dosing schedules, side-effect profiles, and available formulations, which means your oncologist can tailor the choice to your specific circumstances and tolerability. The most commonly reported side effects include a temporary reduction in white blood cell count, known as neutropenia, and fatigue. Both are carefully monitored throughout treatment and are manageable in the majority of patients.
It is worth noting that CDK4/6 inhibitors are taken orally, typically as daily or once-daily tablets, which many women find significantly more compatible with daily life compared to intravenous chemotherapy regimens.
If you have HR+/HER2- metastatic breast cancer and have not yet discussed CDK4/6 inhibitor therapy with your oncologist, or if you were diagnosed before these agents became the standard of care, this is a conversation worth initiating at your next appointment. Ask specifically whether a CDK4/6 inhibitor should be part of your current treatment plan and, if not, why not.
2. Antibody-Drug Conjugates: Precision Delivery That Changes Everything
One of the most exciting categories of cancer treatment to emerge in recent years is the antibody-drug conjugate, commonly referred to as an ADC. The concept is deceptively elegant: take a highly potent chemotherapy drug and chemically attach it to an antibody that specifically recognises and binds to cancer cells. Instead of flooding the entire body with a toxic agent and accepting widespread collateral damage, the drug is delivered directly to the tumour cell, sparing healthy tissue.
Think of conventional chemotherapy as a broad-spectrum blast across an entire room. An ADC is a precision-guided delivery system that finds the target first, then releases its payload.
The most significant ADC currently reshaping outcomes in breast cancer is trastuzumab deruxtecan, which you may encounter under the brand name Enhertu. Originally developed for HER2-positive breast cancer, where it has produced dramatically improved outcomes over earlier-line HER2-targeted therapies, this drug has now achieved something extraordinary: meaningful clinical benefit in a population no one initially anticipated.
For many years, breast cancer was classified in binary terms as either HER2-positive or HER2-negative. However, research revealed that a large proportion of tumours classified as HER2-negative actually express very low levels of HER2 protein, a category now formally designated as HER2-low. This group was previously considered ineligible for HER2-targeted therapy because their expression levels were too low to be actionable with older treatments.
The DESTINY-Breast04 trial changed this entirely. Women with HER2-low metastatic breast cancer who received trastuzumab deruxtecan demonstrated significantly improved progression-free and overall survival compared to the physician’s choice of chemotherapy. This is a paradigm shift in how a substantial proportion of breast cancer patients are classified and treated.
For women with HER2-positive metastatic breast cancer, additional ADCs including trastuzumab emtansine have been available for several years, and newer agents in this class continue to show promise in ongoing trials.
The practical implication for you: ask your oncologist whether your pathology report includes HER2-low status. If your tumour has been classified as HER2-negative but has not been specifically assessed for low-level HER2 expression, request that assessment. It may open a treatment pathway you were previously told was not available to you.
3. PARP Inhibitors: Turning Cancer’s Own Weakness Against It
Approximately 5 to 10 percent of all breast cancers are linked to inherited mutations in the BRCA1 or BRCA2 genes. When functioning normally, these genes are essential guardians of DNA integrity, coordinating repair processes that prevent genetic damage from accumulating and driving abnormal cell growth. When BRCA1 or BRCA2 is mutated, this repair function is compromised, and the cells become significantly more vulnerable to DNA damage.
PARP inhibitors exploit this vulnerability with a mechanism that is, clinically speaking, one of the more elegant strategies in modern oncology.
PARP, which stands for poly ADP-ribose polymerase, is another DNA repair enzyme. In normal cells, PARP provides a backup DNA repair pathway. In cells where BRCA1 or BRCA2 is already non-functional, PARP represents one of the few remaining repair mechanisms. When you inhibit PARP in a BRCA-mutated cancer cell, you remove that last safety net. The cell, already struggling to manage DNA damage with a defective BRCA gene, is unable to repair itself through any available route. The result is a cascade of irreparable DNA breaks, and the cancer cell collapses. This mechanism is known as synthetic lethality, a term that describes the phenomenon whereby two individually non-lethal impairments become lethal when combined.
Clinical trial data, including the OlympiAD study for olaparib and the EMBRACA trial for talazoparib, have demonstrated that PARP inhibitors extend progression-free survival compared to standard single-agent chemotherapy in patients with germline BRCA1 or BRCA2 mutations and HER2-negative metastatic breast cancer. Crucially, these are oral drugs taken daily, which most women find considerably more manageable than intravenous chemotherapy regimens.
If you have a personal or family history of breast cancer, ovarian cancer, or other BRCA-related malignancies and have not yet undergone genetic counselling and testing for BRCA1 and BRCA2 mutations, this is a clinical priority. A positive result is not simply a treatment consideration for you. It is vital information for your first-degree relatives as well. Ask your oncologist for a referral to a genetic counsellor.
4. Immunotherapy: Activating the Body’s Most Sophisticated Defence System
For women with triple-negative breast cancer, immunotherapy has opened a chapter that genuinely did not exist five years ago.
Your immune system is designed to identify and destroy abnormal cells, including those that become cancerous. Many tumours, however, have evolved mechanisms to evade this surveillance, in effect hiding from the immune system by displaying specific molecular signals that tell immune cells to stand down. Checkpoint inhibitors, the most widely used class of cancer immunotherapy, work by blocking these “stand down” signals, thereby allowing the immune system to recognise and mount an attack against the tumour.
The checkpoint inhibitor pembrolizumab, which targets the PD-1 pathway, has been approved in combination with chemotherapy for early-stage, high-risk, PD-L1-positive triple-negative breast cancer. The PD-L1 biomarker indicates whether the tumour is expressing the specific molecular signal that the drug is designed to block. The KEYNOTE-522 trial demonstrated that adding pembrolizumab to chemotherapy significantly improved pathological complete response rates in early TNBC, meaning a meaningfully larger proportion of women showed no detectable cancer in their breast or lymph nodes after treatment. This has significant implications for long-term recurrence risk and overall outcomes.
In the metastatic TNBC setting, ongoing clinical trials are investigating immunotherapy in combination with newer agents including antibody-drug conjugates, with early signals suggesting meaningful additive activity. The field is moving with considerable speed.
It is important to note that immunotherapy is not universally effective across all TNBC. Biomarker testing, specifically testing the tumour for PD-L1 expression, is essential before initiating this class of treatment and should be considered standard of care. If you have TNBC and have not had your tumour assessed for PD-L1 status, ask your oncologist about this test.
5. PI3K and AKT Pathway Inhibitors: Targeting the Resistance Escape Route
One of the most common reasons that HR+/HER2- metastatic breast cancer eventually stops responding to oestrogen-blocking therapy is the activation of an alternative cellular growth pathway: the PI3K/AKT/mTOR signalling cascade. Think of this pathway as a cellular motorway that carries growth signals deep into the nucleus, instructing the cell to continue dividing even when oestrogen is no longer available to fuel it. When this pathway becomes constitutively active, cancer cells effectively bypass the blockade that hormone therapy creates.
The mutation most frequently responsible for this bypass is found in the PIK3CA gene, which is present in approximately 40 percent of HR+/HER2- breast cancers. Alpelisib is a PI3K inhibitor specifically designed to block the protein produced by this mutated gene. The SOLAR-1 trial demonstrated that combining alpelisib with fulvestrant, a standard hormone-blocking drug, significantly improved progression-free survival in patients with PIK3CA-mutated, HR+/HER2- advanced breast cancer who had previously received hormone therapy.
More recently, capivasertib, which targets the AKT protein further downstream within the same signalling pathway, has demonstrated clinical benefit in the CAPItello-291 trial in combination with fulvestrant. The important distinction is that capivasertib shows activity regardless of PIK3CA mutation status, which potentially broadens the population of women who may benefit.
These agents carry specific side effects that require monitoring and proactive management, most notably elevated blood glucose, known as hyperglycaemia, and skin rashes. Your oncology team should discuss these with you in advance and establish a monitoring plan before you begin treatment.
Testing for PIK3CA mutation status, which can be done using either tumour tissue or a liquid biopsy from a blood sample, should now be considered standard of care in the management of HR+/HER2- metastatic breast cancer. If you have not yet been tested for this mutation, ask your oncologist specifically.
6. Oral SERDs and Next-Generation Endocrine Therapy: Beyond the Aromatase Inhibitor Era
When hormone-blocking therapy stops working in HR+/HER2- metastatic breast cancer, one of the most common mechanisms underlying this resistance is the acquisition of mutations in the oestrogen receptor gene itself, known as ESR1 mutations. These mutations alter the shape of the oestrogen receptor in a way that allows it to remain permanently active, even in the complete absence of oestrogen. This is the cancer effectively rewiring its own receptor so that the switch is stuck in the “on” position, regardless of whether the hormone that normally flips the switch is present.
Elacestrant is a next-generation oral selective oestrogen receptor degrader, or SERD, designed specifically to overcome ESR1 mutations. Unlike fulvestrant, which requires monthly intramuscular injections and has limitations in terms of bioavailability, elacestrant is taken as a single daily oral tablet. The EMERALD trial demonstrated that elacestrant significantly improved progression-free survival in women with ESR1-mutated HR+/HER2- metastatic breast cancer who had previously received a CDK4/6 inhibitor plus hormone therapy.
This is clinically meaningful for several reasons. ESR1 mutations do not typically exist at the time of initial diagnosis. They emerge over time as a resistance mechanism following exposure to aromatase inhibitors. Liquid biopsy, described in the following section, can now detect these mutations in circulating tumour DNA before they cause visible disease progression, allowing earlier switching to an agent like elacestrant before the cancer advances.
Several additional oral SERDs are currently in clinical trials at various stages of development, with preliminary data suggesting they may have roles in earlier lines of therapy as well. The shift from injectable to oral endocrine therapy is not simply a convenience improvement. It represents a meaningful quality-of-life advancement for women managing treatment over months and years.
If you are currently receiving fulvestrant after hormone therapy resistance and have not been tested for ESR1 mutations, ask your oncologist about liquid biopsy testing to inform whether an oral SERD might be appropriate for you.
7. Liquid Biopsy and Comprehensive Genomic Profiling: The Power of Knowing More
One of the quieter but genuinely transformative advances in the management of metastatic breast cancer over recent years is the clinical integration of liquid biopsy, a simple blood test that detects fragments of tumour DNA circulating in the bloodstream. This technology analyses what is known as circulating tumour DNA, or ctDNA, and the implications for treatment decision-making are profound.
Consider the limitations of a conventional tissue biopsy. It captures a snapshot of one region of the tumour at one point in time. But tumours are not static. They evolve under the pressure of treatment, developing new mutations as they acquire resistance. A biopsy performed at the time of initial diagnosis may not reflect the molecular landscape of the same tumour two years later, particularly after it has been exposed to several lines of therapy.
Liquid biopsy addresses this limitation elegantly. It can identify specific genetic mutations driving the cancer at any given moment, including PIK3CA mutations and ESR1 mutations, without requiring an invasive tissue procedure. This allows oncologists to track tumour evolution in real time and to make treatment changes based on current molecular data rather than outdated pathology.
Furthermore, liquid biopsy can detect rising ctDNA levels before a tumour becomes visible on a conventional imaging scan. This provides an early warning of disease progression, sometimes weeks or months ahead of radiological evidence, enabling treatment switches to be made proactively rather than reactively.
Comprehensive genomic profiling of tumour tissue using next-generation sequencing adds another layer of precision. This technology can identify rare or unusual mutations that make a cancer eligible for targeted therapies or clinical trials it would not otherwise qualify for. Some of these mutations are present only in a small percentage of breast cancers but are highly actionable with specific drugs already in development or in trials.
As I’ve seen with many patients, those who undergo comprehensive genomic profiling of their metastatic tumour frequently discover treatment options that were simply not on the initial plan. This is not because their original oncology team was uninformed. It is because the technology to identify these opportunities has matured significantly and is now more widely available than it was even three years ago.
If you have not had either a liquid biopsy or comprehensive genomic profiling of your metastatic disease, ask your oncologist directly about accessing these tests. In many healthcare systems, they are now covered by insurance or available through clinical trials, and the information they provide can change the treatment conversation significantly.
8. Integrative Survivorship Care: Living Well, Not Just Living Longer
Perhaps the most underappreciated shift in metastatic breast cancer care over the last five years is not a drug at all. It is a change in philosophy. Metastatic breast cancer is now increasingly managed as a chronic condition to be lived with, rather than treated purely as an acute crisis. This shift has driven meaningful changes in how comprehensive care is delivered.
Bone health management has become a critical and formally integrated component of care, particularly for women with bone metastases or those receiving aromatase inhibitor therapy, which accelerates bone mineral loss. Bone-strengthening agents, including bisphosphonates and RANK-L inhibitors, are now routinely incorporated into treatment plans, where they reduce the risk of skeletal-related events such as fractures. Some analyses have also identified modest anti-tumour effects from these agents, which is an additional consideration when discussing treatment decisions with your oncologist.
Palliative care, which is frequently and incorrectly equated in the public mind with end-of-life care, is now recognised across oncology as a clinical discipline that should be integrated from the point of metastatic diagnosis. Research has consistently demonstrated that early palliative care integration improves quality of life, symptom control, and treatment tolerability. Notably, some well-designed studies have identified a modest overall survival benefit associated with early palliative care involvement, a finding that challenges the assumption that survival and comfort-focused care are competing priorities.
Psychological support has moved from the periphery of metastatic breast cancer care to a recognised clinical necessity. Living with stage 4 breast cancer carries a significant and often underacknowledged psychological burden. Unmanaged anxiety, depression, and post-traumatic stress symptoms impair treatment adherence, disrupt sleep, worsen fatigue, and reduce quality of life in measurable ways. Cognitive behavioural therapy, structured peer support, and, where clinically appropriate, pharmacological support are all part of a comprehensive survivorship approach.
Exercise prescription, nutritional guidance, and sleep support have all been shown in multiple trials to reduce cancer-related fatigue, improve treatment tolerability, and enhance wellbeing in women with metastatic cancer. Exercise in particular, even moderate, regular movement, has been shown to reduce fatigue, the most commonly reported and disabling symptom in this population, and emerging data suggest it may have modest effects on disease biology as well.
According to Mayo Clinic’s comprehensive resource on breast cancer treatment and care, an integrated approach to care that addresses physical wellbeing, emotional health, and symptom management alongside active oncological treatment is associated with better overall outcomes for women with advanced breast cancer. You deserve this standard of care. If survivorship support is not currently part of your treatment plan, ask your oncology team how to access it.
In My 19 Years of Clinical Practice, What I’ve Seen Most Often Is This
In my 19 years of clinical practice, what I’ve seen most often is women arriving at a consultation after a stage 4 diagnosis holding a printed sheet from the internet, reading a median survival figure that makes the room feel very small. And then I have watched those same women, carefully navigated through the landscape of modern treatment, return to clinic five years later talking about a holiday they have just booked, a milestone they are planning for, an ordinary Tuesday that felt like a gift.
The statistics have never captured this. They were never designed to. Statistics describe populations. Medicine, at its best, treats individuals.
What I have consistently observed is that the women who achieve the most meaningful and sustained outcomes are those who are actively engaged in their own care. They ask for genomic profiling. They seek second opinions at specialist centres when something feels incomplete. They ask their oncologist directly whether clinical trials are an option. They take seriously the role of sleep, movement, psychological support, and nutrition alongside their clinical treatment. They do not accept vague answers to specific questions.
I have also seen, more times than I would like, the cost of care that is not kept up to date with current evidence. New treatments receive approval with increasing frequency. Academic cancer centres and specialist metastatic breast cancer clinics have access to emerging therapies and clinical trial networks that general oncology practices may not. A second opinion from a centre that specialises in metastatic breast cancer is not an act of disloyalty toward your current team. It is a clinical imperative. The women who pursue them often come back with new options on the table.
You are entitled to the best available care. Do not accept less.
When to See a Specialist: Red Flags That Warrant Urgent Attention
Metastatic breast cancer requires expert, ongoing management. But there are specific circumstances in which you should not wait until your next scheduled appointment.
Bone pain that develops suddenly or worsens significantly over one to two weeks, particularly in the spine or hips, warrants an urgent call to your oncology team the same day. Bone metastases can predispose to fractures, and involvement of the spinal cord, while uncommon, can cause neurological damage if not addressed promptly. Describe the location, severity, and rate of onset clearly when you call.
New or rapidly worsening headaches, visual disturbances, confusion, difficulties with balance, or coordination problems may indicate brain metastases. Contact your oncology team the same day or go to your nearest emergency department. Brain metastases are treatable, particularly with stereotactic radiosurgery, but they require prompt imaging and evaluation.
Significant new shortness of breath may suggest a pleural effusion, which is fluid accumulating around the lung, or disease involvement of the lungs themselves. This warrants urgent review, ideally the same day, either with your oncology team or through your emergency department.
Signs of liver involvement, including yellowing of the skin or the whites of the eyes (jaundice), sudden and significant reduction in appetite, or rapid abdominal distension, should prompt contact with your oncology team within 24 to 48 hours. These symptoms may indicate hepatic disease progression requiring prompt reassessment of your treatment plan.
Any acute neurological change, including sudden weakness affecting one side of the body, speech difficulties, or severe and unexplained dizziness, requires emergency medical attention regardless of the time of day or night.
Beyond acute symptoms, if you feel that your current treatment team is not offering you access to current genomic profiling, clinical trial participation, or up-to-date targeted therapies, this is a legitimate and important reason to seek a second opinion at a specialist cancer centre or academic medical institution. Ask your oncologist for a referral to a metastatic breast cancer specialist or request a multidisciplinary tumour board review of your case. You are entitled to this.
You Are Not a Statistic: Moving Forward with Hope and Clarity
If this article gives you one thing to hold onto, let it be this: the stage 4 breast cancer life expectancy data you encounter in 2026 does not tell the full story of what is possible under current treatment.
CDK4/6 inhibitors have transformed hormone receptor-positive metastatic breast cancer into a condition where years of progression-free life are a realistic expectation for many women. Antibody-drug conjugates have opened doors for HER2-low patients who were previously told targeted therapy was not an option for them. PARP inhibitors offer precision medicine for BRCA mutation carriers. Immunotherapy has redrawn the map for triple-negative breast cancer. Liquid biopsy and genomic profiling are making treatment decisions more precise and more personalised than at any point in the history of oncology. And integrative survivorship care is ensuring that women do not simply live longer, but live better.
According to the National Cancer Institute’s patient guide to breast cancer treatment, precision medicine approaches based on the specific molecular characteristics of a tumour are now central to the standard of care for metastatic breast cancer. You deserve access to this level of individualised treatment.
Your most concrete next step is this: if you have not yet had comprehensive genomic profiling of your metastatic tumour, bring it up at your next appointment. Ask your oncologist what the results would tell you, which treatment decisions it would inform, and how to access the test if it is not already scheduled. That conversation may be the most important one you have this year.
Share this article with someone who needs it tonight. A diagnosis like this is rarely carried alone, and the people who love you deserve to understand what is possible too.
Drop a comment below and tell us what has helped you most. Your story matters more than you know. It may be exactly the thread of hope that someone else is searching for at 2:47 in the morning.
Medical Disclaimer: This article is for informational purposes only and does not constitute medical advice. Always consult a qualified healthcare provider before making any changes to your health or treatment plan.